Three dimensional electrospun PCL/PLA blend nanofibrous scaffolds with significantly improved stem cells osteogenic differentiation and cranial bone formation
- PMID: 27886552
- PMCID: PMC5181114
- DOI: 10.1016/j.biomaterials.2016.11.018
Three dimensional electrospun PCL/PLA blend nanofibrous scaffolds with significantly improved stem cells osteogenic differentiation and cranial bone formation
Abstract
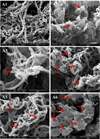
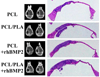
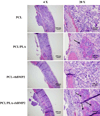
Nanofibrous scaffolds that are morphologically/structurally similar to natural ECM are highly interested for tissue engineering; however, the electrospinning technique has the difficulty in directly producing clinically relevant 3D nanofibrous scaffolds with desired structural properties. To address this challenge, we have developed an innovative technique of thermally induced nanofiber self-agglomeration (TISA) recently. The aim of this work was to prepare (via the TISA technique) and evaluate 3D electrospun PCL/PLA blend (mass ratio: 4/1) nanofibrous scaffolds having high porosity of ∼95.8% as well as interconnected and hierarchically structured pores with sizes from sub-micrometers to ∼300 μm for bone tissue engineering. The hypothesis was that the incorporation of PLA (with higher mechanical stiffness/modulus and bioactivity) into PCL nanofibers would significantly improve human mesenchymal stem cells (hMSCs) osteogenic differentiation in vitro and bone formation in vivo. Compared to neat PCL-3D scaffolds, PCL/PLA-3D blend scaffolds had higher mechanical properties and in vitro bioactivity; as a result, they not only enhanced the cell viability of hMSCs but also promoted the osteogenic differentiation. Furthermore, our in vivo studies revealed that PCL/PLA-3D scaffolds considerably facilitated new bone formation in a critical-sized cranial bone defect mouse model. In summary, both in vitro and in vivo results indicated that novel 3D electrospun PCL/PLA blend nanofibrous scaffolds would be strongly favorable/desired for hMSCs osteogenic differentiation and cranial bone formation.
Keywords: 3D electrospun nanofibrous scaffold; Bone regeneration; Osteogenic differentiation; Polycaprolactone; Polylactic acid.
Copyright © 2016 Elsevier Ltd. All rights reserved.
Figures







References
-
- Cabraja M, Kroppenstedt S. Bone grafting and substitutes in spine surgery. J Neurosurg Sci. 2012;56:87–95. - PubMed
-
- Morelli S, Salerno S, Holopainen J, Ritala M, De Bartolo L. Osteogenic and osteoclastogenic differentiation of co-cultured cells in polylactic acid–nanohydroxyapatite fiber scaffolds. J Biotechnol. 2015;204:53–62. - PubMed
-
- Heymer A, Bradica G, Eulert J, Nöth U. Multiphasic collagen fibre–PLA composites seeded with human mesenchymal stem cells for osteochondral defect repair: an in vitro study. J Tissue Eng Regen Med. 2009;3:389–397. - PubMed
-
- Khan Y, Yaszemski MJ, Mikos AG, Laurencin CT. Tissue engineering of bone: material and matrix considerations. J Bone Joint Surg. 2008;90:36–42. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

