Robot-assisted mechanical therapy attenuates stroke-induced limb skeletal muscle injury
- PMID: 27895105
- PMCID: PMC5295731
- DOI: 10.1096/fj.201600437R
Robot-assisted mechanical therapy attenuates stroke-induced limb skeletal muscle injury
Abstract
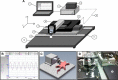
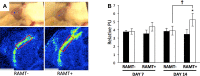
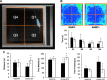
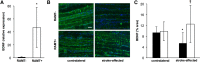
The efficacy and optimization of poststroke physical therapy paradigms is challenged in part by a lack of objective tools available to researchers for systematic preclinical testing. This work represents a maiden effort to develop a robot-assisted mechanical therapy (RAMT) device to objectively address the significance of mechanical physiotherapy on poststroke outcomes. Wistar rats were subjected to right hemisphere middle-cerebral artery occlusion and reperfusion. After 24 h, rats were split into control (RAMT-) or RAMT+ groups (30 min daily RAMT over the stroke-affected gastrocnemius) and were followed up to poststroke d 14. RAMT+ increased perfusion 1.5-fold in stroke-affected gastrocnemius as compared to RAMT- controls. Furthermore, RAMT+ rats demonstrated improved poststroke track width (11% wider), stride length (21% longer), and travel distance (61% greater), as objectively measured using software-automated testing platforms. Stroke injury acutely increased myostatin (3-fold) and lowered brain-derived neurotrophic factor (BDNF) expression (0.6-fold) in the stroke-affected gastrocnemius, as compared to the contralateral one. RAMT attenuated the stroke-induced increase in myostatin and increased BDNF expression in skeletal muscle. Additional RAMT-sensitive myokine targets in skeletal muscle (IL-1ra and IP-10/CXCL10) were identified from a cytokine array. Taken together, outcomes suggest stroke acutely influences signal transduction in hindlimb skeletal muscle. Regimens based on mechanical therapy have the clear potential to protect hindlimb function from such adverse influence.-Sen, C. K., Khanna, S., Harris, H., Stewart, R., Balch, M., Heigel, M., Teplitsky, S., Gnyawali, S., Rink, C. Robot-assisted mechanical therapy attenuates stroke-induced limb skeletal muscle injury.
Keywords: cerebrovascular accident; ischemia; physical therapy; rehabilitation.
© FASEB.
Figures



References
-
- American Heart Association Council on Cardiovascular Nursing and the Stroke Council (2010) Comprehensive overview of nursing and interdisciplinary rehabilitation care of the stroke patient: a scientific statement from the American Heart Association. Stroke 41, 2402–2448 - PubMed
-
- Langhorne P., Bernhardt J., Kwakkel G. (2011) Stroke rehabilitation. Lancet 377, 1693–1702 - PubMed
-
- Lennon S., Baxter D., Ashburn A. (2001) Physiotherapy based on the Bobath concept in stroke rehabilitation: a survey within the UK. Disabil. Rehabil. 23, 254–262 - PubMed
-
- Kollen B. J., Lennon S., Lyons B., Wheatley-Smith L., Scheper M., Buurke J. H., Halfens J., Geurts A. C., Kwakkel G. (2009) The effectiveness of the Bobath concept in stroke rehabilitation: what is the evidence? Stroke 40, e89–e97 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

