Cytokines and Cytokine Receptors Involved in the Pathogenesis of Alzheimer's Disease
- PMID: 27895978
- PMCID: PMC5123596
- DOI: 10.4172/2155-9899.1000441
Cytokines and Cytokine Receptors Involved in the Pathogenesis of Alzheimer's Disease
Abstract
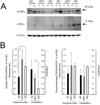
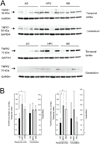
Inflammatory mechanisms are implicated in the pathology of Alzheimer's disease (AD). However, it is unclear whether inflammatory alterations are a cause or consequence of neurodegeneration leading to dementia. Clarifying this issue would provide valuable insight into the early diagnosis and therapeutic management of AD. To address this, we compared the mRNA expression profiles of cytokines in the brains of AD patients with "non-demented individuals with AD pathology" and non-demented healthy control (ND) individuals. "Non-demented individuals with AD pathology" are referred to as high pathology control (HPC) individuals that are considered an intermediate subset between AD and ND. HPC represents a transition between normal aging and early stage of AD, and therefore, is useful for determining whether neuroinflammation is a cause or consequence of AD pathology. We observed that immunological conditions that produce cytokines in the HPC brain were more representative of ND than AD. To validate these result, we investigated the expression of inflammatory mediators at the protein level in postmortem brain tissues. We examined the protein expression of tumor necrosis factor (TNF)α and its receptors (TNFRs) in the brains of AD, HPC, and ND individuals. We found differences in soluble TNFα and TNFRs expression between AD and ND groups and between AD and HPC groups. Expression in the temporal cortex was lower in the AD brains than HPC and ND. Our findings indicate that alterations in immunological conditions involving TNFR-mediated signaling are not the primary events initiating AD pathology, such as amyloid plaques and tangle formation. These may be early events occurring along with synaptic and neuronal changes or later events caused by these changes. In this review, we emphasize that elucidating the temporal expression of TNFα signaling molecules during AD is important to understand the selective tuning of these pathways required to develop effective therapeutic strategies for AD.
Keywords: Alzheimer’s disease; Cytokines; Neuroinflammation; Postmortem brain tissues; TNF receptors; Tumor necrosis factor (TNF)α.
Figures
Similar articles
-
Expression profiles of cytokines in the brains of Alzheimer's disease (AD) patients compared to the brains of non-demented patients with and without increasing AD pathology.J Alzheimers Dis. 2011;25(1):59-76. doi: 10.3233/JAD-2011-101815. J Alzheimers Dis. 2011. PMID: 21368376 Free PMC article.
-
Multiple cytokines are involved in the early events leading to the Alzheimer's disease pathology.Tottori Rinsho Kagaku Kenkyukai Shi. 2008;1(2):359-373. Tottori Rinsho Kagaku Kenkyukai Shi. 2008. PMID: 22586434 Free PMC article.
-
Alzheimer's disease and non-demented high pathology control nonagenarians: comparing and contrasting the biochemistry of cognitively successful aging.PLoS One. 2011;6(11):e27291. doi: 10.1371/journal.pone.0027291. Epub 2011 Nov 7. PLoS One. 2011. PMID: 22087282 Free PMC article.
-
Modeling microglial activation in Alzheimer's disease with human postmortem microglial cultures.Neurobiol Aging. 2001 Nov-Dec;22(6):945-56. doi: 10.1016/s0197-4580(01)00311-6. Neurobiol Aging. 2001. PMID: 11755003 Review.
-
Role of pro-inflammatory cytokines released from microglia in Alzheimer's disease.Ann Transl Med. 2015 Jun;3(10):136. doi: 10.3978/j.issn.2305-5839.2015.03.49. Ann Transl Med. 2015. PMID: 26207229 Free PMC article. Review.
Cited by
-
Global Gene Expression Profiling and Transcription Factor Network Analysis of Cognitive Aging in Monozygotic Twins.Front Genet. 2021 Jun 14;12:675587. doi: 10.3389/fgene.2021.675587. eCollection 2021. Front Genet. 2021. PMID: 34194475 Free PMC article.
-
Alzheimer's disease is an inherent, natural part of human brain aging: an integrated perspective.Free Neuropathol. 2022 Jul 8;3:17. doi: 10.17879/freeneuropathology-2022-3806. eCollection 2022 Jan. Free Neuropathol. 2022. PMID: 37284149 Free PMC article.
-
Protective effect of ISO‑1 against advanced glycation end product aggravation of PC12 cell injury induced by Aβ1‑40.Mol Med Rep. 2019 Sep;20(3):2135-2142. doi: 10.3892/mmr.2019.10483. Epub 2019 Jul 9. Mol Med Rep. 2019. PMID: 31322215 Free PMC article.
-
Neurodegenerative diseases: model organisms, pathology and autophagy.J Genet. 2018 Jul;97(3):679-701. J Genet. 2018. PMID: 30027903 Review.
-
Microarray Analysis Revealed Inflammatory Transcriptomic Changes after LSL60101 Treatment in 5XFAD Mice Model.Genes (Basel). 2021 Aug 26;12(9):1315. doi: 10.3390/genes12091315. Genes (Basel). 2021. PMID: 34573297 Free PMC article.
References
-
- Eikelenboom P, Bate C, Van Gool WA, Hoozemans JJ, Rozemuller JM, et al. Neuroinflammation in Alzheimer’s disease and prion disease. Glia. 2002;40:232–239. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
