LEAPing through the looking glass: secondary analysis of the effect of skin test size and age of introduction on peanut tolerance after early peanut introduction
- PMID: 27896827
- PMCID: PMC5447487
- DOI: 10.1111/all.13100
LEAPing through the looking glass: secondary analysis of the effect of skin test size and age of introduction on peanut tolerance after early peanut introduction
Abstract
Background: In the Learning Early About Peanut Allergy (LEAP) study, early peanut introduction in high-risk 4- to 11-month-olds was associated with a significantly decreased risk of developing peanut allergy. However, the influences of key baseline high-risk factors on peanut tolerance are poorly understood.
Methods: Secondary analysis was conducted on the publically available LEAP dataset, exploring relationships between peanut tolerance, baseline peanut/egg sensitization, eczema severity/duration, age of introduction, gender, and race.
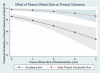
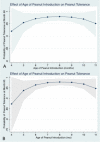
Results: A multiple logistic regression model predicting odds of successful oral food challenge (OFC) at 60 months noted higher odds with early introduction (OR 9.2, P < 0.001, 95% CI 4.2-20.3), white race (OR 2.1, P = 0.04, 95% CI 1.1-3.9), and advancing age (OR 4.8, P = 0.04, 95% CI 1.1-20.8). Odds of peanut tolerance were lower with increasing peanut wheal size (OR 0.58, P < 0.001, 95% CI 0.46-0.74), increased baseline SCORAD score (OR 0.98, P = 0.04, 95% CI 0.97-1), and increased kUA /l of egg serum IgE (sIgE) (OR 0.99, P = 0.04, 95% CI 0.98-1). The probability of peanut tolerance in the early introduction group was 83% vs 43% in the avoidance group with SPT wheal of <4 mm. The probability of a successful OFC was significantly higher with peanut introduction between 6 and 11 months than at 4-6 months. Increasing eczema severity had limited impact on the probability of peanut tolerance in the early introduction arm.
Conclusion: Increasing peanut wheal size predicted peanut tolerance only in the avoidance arm. Peanut introduction between 6 and 11 months of age was associated with the highest rates of peanut tolerance, questioning the 'urgency' of introduction before 6 months.
Keywords: Learning Early about Peanut; allergy prevention; complementary feeding; early peanut introduction; peanut allergy.
© 2016 John Wiley & Sons A/S. Published by John Wiley & Sons Ltd.
Figures
Comment in
-
Letter of response to Greenhawt et al. 'LEAPing Through the Looking Glass: Secondary Analysis of the Effect of Skin Test Size and Age of Introduction on Peanut Tolerance after Early Peanut Introduction'.Allergy. 2017 Aug;72(8):1267-1271. doi: 10.1111/all.13127. Allergy. 2017. PMID: 28691223 Free PMC article. No abstract available.
References
-
- Gupta RS, Springston EE, Warrier MR, Smith B, Kumar R, Pongracic J, et al. The prevalence, severity, and distribution of childhood food allergy in the United States. Pediatrics. 2011;128:e9–17. - PubMed
-
- Osborne NJ, Koplin JJ, Martin PE, Gurrin LC, Lowe AJ, Matheson MC, et al. Prevalence of challenge-proven IgE-mediated food allergy using population-based sampling and predetermined challenge criteria in infants. J Allergy Clin Immunol. 2011;127:668–76. e1–2. - PubMed
-
- Sampson HA, Aceves S, Bock SA, James J, Jones S, Lang D, et al. Food allergy: a practice parameter update-2014. J Allergy Clin Immunol. 2014;134:1016–25. e43. - PubMed
-
- Sicherer SH, Munoz-Furlong A, Godbold JH, Sampson HA. US prevalence of self-reported peanut, tree nut, and sesame allergy: 11-year follow-up. J Allergy Clin Immunol. 2010;125:1322–6. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

