Preparation of 6/8/11-Amino/Chloro-Oxoisoaporphine and Group-10 Metal Complexes and Evaluation of Their in Vitro and in Vivo Antitumor Activity
- PMID: 27898051
- PMCID: PMC5127189
- DOI: 10.1038/srep37644
Preparation of 6/8/11-Amino/Chloro-Oxoisoaporphine and Group-10 Metal Complexes and Evaluation of Their in Vitro and in Vivo Antitumor Activity
Abstract
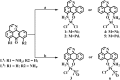
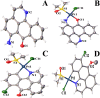
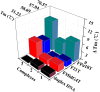
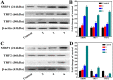
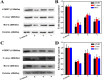
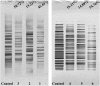
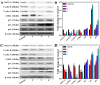
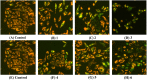
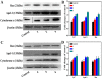
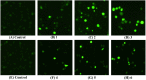
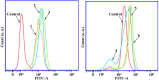
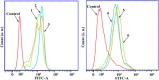
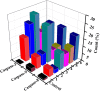
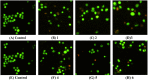
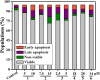
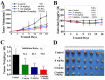
A series of group-10 metal complexes 1-14 of oxoisoaporphine derivatives were designed and synthesized. 1-14 were more selectively cytotoxic to Hep-G2 cells comparing with normal liver cells. In vitro cytotoxicity results showed that complexes 1-6, 7, 8, 10 and 11, especially 3, were telomerase inhibitors targeting c-myc, telomeric, and bcl-2 G4s and triggered cell senescence and apoptosis; they also caused telomere/DNA damage and S phase arrest. In addition, 1-6 also caused mitochondrial dysfunction. Notably, 3 with 6-amino substituted ligand La exhibited less side effects than 6 with 8-amino substituted ligand Lb and cisplatin, but similar tumor growth inhibition efficacy in BEL-7402 xenograft model. Complex 3 has the potential to be developed as an effective anticancer agent.
Figures












Similar articles
-
High in vivo antitumor activity of cobalt oxoisoaporphine complexes by targeting G-quadruplex DNA, telomerase and disrupting mitochondrial functions.Eur J Med Chem. 2016 Nov 29;124:380-392. doi: 10.1016/j.ejmech.2016.08.063. Epub 2016 Aug 28. Eur J Med Chem. 2016. PMID: 27597414
-
5-Bromo-oxoisoaporphine platinum(II) complexes exhibit tumor cell cytotoxcicity via inhibition of telomerase activity and disruption of c-myc G-quadruplex DNA and mitochondrial functions.Eur J Med Chem. 2018 Feb 10;145:360-369. doi: 10.1016/j.ejmech.2017.12.092. Epub 2018 Jan 4. Eur J Med Chem. 2018. PMID: 29335202
-
Water-Soluble Ruthenium(II) Complexes with Chiral 4-(2,3-Dihydroxypropyl)-formamide Oxoaporphine (FOA): In Vitro and in Vivo Anticancer Activity by Stabilization of G-Quadruplex DNA, Inhibition of Telomerase Activity, and Induction of Tumor Cell Apoptosis.J Med Chem. 2015 Jun 11;58(11):4771-89. doi: 10.1021/acs.jmedchem.5b00444. Epub 2015 Jun 2. J Med Chem. 2015. PMID: 25988535
-
Ruthenium(II) Complexes as Potential Apoptosis Inducers in Chemotherapy.Anticancer Agents Med Chem. 2017;17(1):29-39. Anticancer Agents Med Chem. 2017. PMID: 27349451 Review.
-
Nucleic Acid Quadruplexes and Metallo-Drugs.Met Ions Life Sci. 2018 Feb 5;18:/books/9783110470734/9783110470734-018/9783110470734-018.xml. doi: 10.1515/9783110470734-018. Met Ions Life Sci. 2018. PMID: 29394031 Review.
Cited by
-
Facile total synthesis of lysicamine and the anticancer activities of the RuII, RhIII, MnII and ZnII complexes of lysicamine.Oncotarget. 2017 Jul 26;8(35):59359-59375. doi: 10.18632/oncotarget.19584. eCollection 2017 Aug 29. Oncotarget. 2017. PMID: 28938642 Free PMC article.
-
Oxoisoaporphines and Aporphines: Versatile Molecules with Anticancer Effects.Molecules. 2019 Dec 27;25(1):108. doi: 10.3390/molecules25010108. Molecules. 2019. PMID: 31892146 Free PMC article. Review.
-
Synthesis, characterization and biological evaluation of six highly cytotoxic ruthenium(ii) complexes with 4'-substituted-2,2':6',2''-terpyridine.Medchemcomm. 2018 Feb 2;9(3):525-533. doi: 10.1039/c7md00532f. eCollection 2018 Mar 1. Medchemcomm. 2018. PMID: 30108943 Free PMC article.
-
Oxoaporphine Metal Complexes (CoII, NiII, ZnII) with High Antitumor Activity by Inducing Mitochondria-Mediated Apoptosis and S-phase Arrest in HepG2.Sci Rep. 2017 Apr 24;7:46056. doi: 10.1038/srep46056. Sci Rep. 2017. PMID: 28436418 Free PMC article.
-
4-Hydroxyl-oxoisoaporphine, one small molecule as theranostic agent for simultaneous fluorescence imaging and photodynamic therapy as type II photosensitizer.Photochem Photobiol Sci. 2021 Apr;20(4):501-512. doi: 10.1007/s43630-021-00030-0. Epub 2021 Mar 20. Photochem Photobiol Sci. 2021. PMID: 33743176
References
-
- Han H. & Hurley L. H. G-Quadruplex DNA: a potential target for anti-cancer drug design. Trends Pharmacol. Sci. 21, 136–142 (2000). - PubMed
-
- Amrane S. et al. Topology of a NDA G-qudruplex structure formed in the HIV-1 promoter: a potential target for anti-HIV drug development. J. Am. Chem. Soc. 136, 5249–5252 (2014). - PubMed
-
- Paritala H. & Firestine S. M. Characterization of insulin ILPR sequences for their ability to adopt a G-quadruplex structure. Nucleos. Nucleot. Nucl. 29, 81–90 (2010). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

