Evaluation of the Activity of Lamivudine and Zidovudine against Ebola Virus
- PMID: 27902714
- PMCID: PMC5130197
- DOI: 10.1371/journal.pone.0166318
Evaluation of the Activity of Lamivudine and Zidovudine against Ebola Virus
Abstract
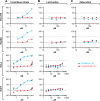
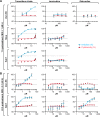
In the fall of 2014, an international news agency reported that patients suffering from Ebola virus disease (EVD) in Liberia were treated successfully with lamivudine, an antiviral drug used to treat human immunodeficiency virus-1 and hepatitis B virus infections. According to the report, 13 out of 15 patients treated with lamivudine survived and were declared free from Ebola virus disease. In this study, the anti-Ebola virus (EBOV) activity of lamivudine and another antiretroviral, zidovudine, were evaluated in a diverse set of cell lines against two variants of wild-type EBOV. Variable assay parameters were assessed to include different multiplicities of infection, lengths of inoculation times, and durations of dosing. At a multiplicity of infection of 1, lamivudine and zidovudine had no effect on EBOV propagation in Vero E6, Hep G2, or HeLa cells, or in primary human monocyte-derived macrophages. At a multiplicity of infection of 0.1, zidovudine demonstrated limited anti-EBOV activity in Huh 7 cells. Under certain conditions, lamivudine had low anti-EBOV activity at the maximum concentration tested (320 μM). However, lamivudine never achieved greater than 30% viral inhibition, and the activity was not consistently reproducible. Combination of lamivudine and zidovudine showed no synergistic antiviral activity. Independently, a set of in vitro experiments testing lamivudine and zidovudine for antiviral activity against an Ebola-enhanced green fluorescent protein reporter virus was performed at the Centers for Disease Control and Prevention. No antiviral activity was observed for either compound. A study evaluating the efficacy of lamivudine in a guinea pig model of EVD found no survival benefit. This lack of benefit was observed despite plasma lamivudine concentrations in guinea pig of about 4 μg/ml obtained in a separately conducted pharmacokinetics study. These studies found no evidence to support the therapeutic use of lamivudine for the treatment of EVD.
Conflict of interest statement
The SRI international affiliation does not alter our adherence to PLOS ONE policies on sharing data and materials.
Figures


References
-
- Feldmann H, Slenczka W, Klenk HD. Emerging and reemerging of filoviruses. Arch Virol Suppl. 1996;11:77–100. - PubMed
-
- Georges AJ, Baize S, Leroy EM, Georges-Courbot MC. [Ebola virus: what the practitioner needs to know]. Med Trop (Mars). 1998;58(2):177–86. - PubMed
-
- World Health Organization. Ebola situation report—10 June 2016. Geneva, CH: World Health Organization; 2016. Available: http://apps.who.int/iris/bitstream/10665/208883/1/ebolasitrep_10Jun2016_....
-
- Grygorczuk S, Hermanowska-Szpakowicz T. [Viral hemorrhagic fevers as a biological weapon]. Pol Merkur Lekarski. 2003;14(80):146–9. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

