Expression analysis and clinical significance of eIF4E, VEGF-C, E-cadherin and MMP-2 in colorectal adenocarcinoma
- PMID: 27907907
- PMCID: PMC5356753
- DOI: 10.18632/oncotarget.13453
Expression analysis and clinical significance of eIF4E, VEGF-C, E-cadherin and MMP-2 in colorectal adenocarcinoma
Abstract
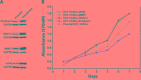
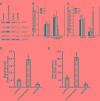
The underlying mechanisms of colorectal carcinoma (CRC) metastasis remain to be elucidated. The aim of this study is to investigate clinical significance and the expression of eIF4E, VEGF-C, MMP-2, and E-cadherin in the CRC metastasis. We investigated their expressions in 108 patients, analyzed the relationships between their expressions in CRC and evaluated the relationships between their expressions and clinical pathogenic parameters. Furthermore, their roles in patient survival and in CRC metastasis were also investigated. We found that eIF4E, VEGF-C and MMP-2 were up-regulated in CRC, and their expression frequencies (EFs) were higher in cancerous tissues than in adjacent normal tissues. The EF of E-cadherin is lower in cancerous tissues than in adjacent normal tissues. Totally, their EFs were not associated with sex and age of patient, however, their EFs were associated with tumor differentiation, the depth of invasion, lymph node metastasis and tumor stages. Furthermore, eIF4E, VEGF-C, and MMP-2 shortened and E-cadherin prolonged survival in patient-derived CRC xenografts. Similarly, eIF4E, VEGF-C, and MMP-2 promoted and E-cadherin suppressed the lung metastasis of CRC cells. In addition, knockdown of eIF4E inhibited migration of CRC cells, downregulated VEGF-C, MMP-2 and upregulated E-cadherin. In conclusion, eIF4E promoted CRC metastasis via up-regulating the expression of VEGF-C, MMP-2 and suppressing E-cadherin.
Keywords: E-cadherin; MMP-2; VEGF-C; colorectal cancer; eIF4E.
Conflict of interest statement
The authors declare no conflicts interest.
Figures




References
-
- American Cancer Association. Cancer Facts & Figures 2016. American Cancer Association.
-
- Sunavala-Dossabhoy G, Palaniyandi S, Clark C, Nathan CO, Abreo FW, Caldito G. Analysis of eIF4E and 4EBP1 mRNAs in head and neck cancer. The Laryngoscope. 2011;121:2136–2141. - PubMed
-
- de Oliveira AT, Reis RM, Afonso J, Martinho O, Matos D, Carvalho AL, Vazquez VL, Silva TB, Scapulatempo C, Saad SS, Longatto-Filho A. Lymphangiogenic VEGF-C and VEGFR-3 expression in genetically characterised gastrointestinal stromal tumours. Histology and histopathology. 2011;26:1499–1507. - PubMed
-
- Beavon IR. The E-cadherin-catenin complex in tumour metastasis: structure, function and regulation. European journal of cancer. 2000;36:1607–1620. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

