Donor-Specific Regulatory T Cells Acquired from Tolerant Mice Bearing Cardiac Allograft Promote Mixed Chimerism and Prolong Intestinal Allograft Survival
- PMID: 27909438
- PMCID: PMC5113131
- DOI: 10.3389/fimmu.2016.00511
Donor-Specific Regulatory T Cells Acquired from Tolerant Mice Bearing Cardiac Allograft Promote Mixed Chimerism and Prolong Intestinal Allograft Survival
Abstract
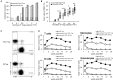
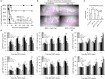
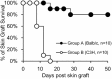
The induction of donor-specific transplant tolerance has always been a central problem for small bowel transplantation (SBT), which is thought to be the best therapy for end-stage bowel failure. With the development of new tolerance-inducing strategies, mixed chimerism induced by co-stimulation blockade has become most potent for tolerance of allografts, such as skin, kidney, and heart. However, a lack of clinically available co-stimulation blockers has hindered efficient application in humans. Furthermore, unlike those for other types of solid organ transplantation, strategies to induce robust mixed chimerism for intestinal allografts have not been fully developed. To improve current mixed chimerism induction protocols for future clinical application, we developed a new protocol using donor-specific regulatory T (Treg) cells from mice with heart allograft tolerance, immunosuppressive drugs which could be used clinically and low doses of irradiation. Our results demonstrated that donor-specific Treg cells acquired from tolerant mice after in vitro expansion generate stable chimerism and lead to acceptance of intestinal allograft. Increased intragraft Treg cells and clonal deletion contribute to the development of SBT tolerance.
Keywords: bone marrow transplantation; donor-specific regulatory T cells; mixed chimerism; small bowel transplantation; transplantation tolerance.
Figures


Similar articles
-
Methyl-Guanine-Methyl-Transferase Transgenic Bone Marrow Transplantation Allows N,N-bis(2-chloroethyl)-Nitrosourea Driven Donor Mixed-Chimerism Without Graft-Versus-Host Disease, and With Donor-Specific Allograft Tolerance.Transplantation. 2015 Dec;99(12):2476-84. doi: 10.1097/TP.0000000000000825. Transplantation. 2015. PMID: 26177088
-
Treg-based therapy and mixed chimerism in small intestinal transplantation: does Treg +BMT equal intestine allograft tolerance?Med Hypotheses. 2011 Jan;76(1):77-8. doi: 10.1016/j.mehy.2010.08.035. Epub 2010 Sep 9. Med Hypotheses. 2011. PMID: 20826066
-
Infusion of Lin- bone marrow cells results in multilineage macrochimerism and skin allograft tolerance in minimally conditioned recipient mice.Transpl Immunol. 2010 Oct;24(1):69-75. doi: 10.1016/j.trim.2010.10.003. Epub 2010 Oct 13. Transpl Immunol. 2010. PMID: 20950686
-
Chimerism studies as an approach for the induction of tolerance to extremity allografts.J Plast Reconstr Aesthet Surg. 2008 Sep;61(9):1009-15. doi: 10.1016/j.bjps.2007.12.082. Epub 2008 Jun 24. J Plast Reconstr Aesthet Surg. 2008. PMID: 18573704 Review.
-
Chimerism and tolerance: past, present and future strategies to prolong renal allograft survival.Curr Opin Nephrol Hypertens. 2021 Jan;30(1):63-74. doi: 10.1097/MNH.0000000000000666. Curr Opin Nephrol Hypertens. 2021. PMID: 33186221 Review.
Cited by
-
Dexamethasone-Induced Myeloid-Derived Suppressor Cells Prolong Allo Cardiac Graft Survival through iNOS- and Glucocorticoid Receptor-Dependent Mechanism.Front Immunol. 2018 Feb 15;9:282. doi: 10.3389/fimmu.2018.00282. eCollection 2018. Front Immunol. 2018. PMID: 29497426 Free PMC article.
-
Adipose-derived mesenchymal stem cells suppress of acute rejection in small bowel transplantation.Saudi J Gastroenterol. 2017 Nov-Dec;23(6):323-329. doi: 10.4103/sjg.SJG_122_17. Saudi J Gastroenterol. 2017. PMID: 29205184 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Other Literature Sources

