IL-9-producing cells in the development of IgE-mediated food allergy
- PMID: 27909880
- PMCID: PMC5225002
- DOI: 10.1007/s00281-016-0605-x
IL-9-producing cells in the development of IgE-mediated food allergy
Abstract
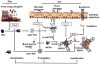
Food allergy is a harmful immune reaction driven by uncontrolled type 2 immune responses. Considerable evidence demonstrates the key roles of mast cells, IgE, and TH2 cytokines in mediating food allergy. However, this evidence provides limited insight into why only some, rather than all, food allergic individuals are prone to develop life-threatening anaphylaxis. Clinical observations suggest that patients sensitized to food through the skin early in life may later develop severe food allergies. Aberrant epidermal thymic stromal lymphopoietin and interleukin (IL) 33 production and genetic predisposition can initiate an allergic immune response mediated by dendritic cells and CD4+TH2 cells in inflamed skin. After allergic sensitization, intestinal IL-25 and food ingestion enhance concerted interactions between type 2 innate lymphoid cells (ILC2s) and CD4+TH2 cells, which perpetuate allergic reactions from the skin to the gut. IL-4 and cross-linking of antigen/IgE/FcεR complexes induce emigrated mast cell progenitors to develop into the multi-functional IL-9-producing mucosal mast cells, which produce prodigious amounts of IL-9 and mast cell mediators to drive intestinal mastocytosis in an autocrine loop. ILC2s and TH9 cells may also serve as alternative cellular sources of IL-9 to augment the amplification of intestinal mastocytosis, which is the key cellular checkpoint in developing systemic anaphylaxis. These findings provide a plausible view of how food allergy develops and progresses in a stepwise manner and that atopic signals, dietary allergen ingestion, and inflammatory cues are fundamental in promoting life-threatening anaphylaxis. This information will aid in improving diagnosis and developing more effective therapies for food allergy-triggered anaphylaxis.
Figures
Similar articles
-
Developing food allergy: a potential immunologic pathway linking skin barrier to gut.F1000Res. 2016 Nov 10;5:F1000 Faculty Rev-2660. doi: 10.12688/f1000research.9497.1. eCollection 2016. F1000Res. 2016. PMID: 27853507 Free PMC article. Review.
-
Induction of Interleukin-9-Producing Mucosal Mast Cells Promotes Susceptibility to IgE-Mediated Experimental Food Allergy.Immunity. 2015 Oct 20;43(4):788-802. doi: 10.1016/j.immuni.2015.08.020. Epub 2015 Sep 22. Immunity. 2015. PMID: 26410628 Free PMC article.
-
IgE and IgG Antibodies as Regulators of Mast Cell and Basophil Functions in Food Allergy.Front Immunol. 2020 Dec 11;11:603050. doi: 10.3389/fimmu.2020.603050. eCollection 2020. Front Immunol. 2020. PMID: 33362785 Free PMC article. Review.
-
Basophil-derived IL-4 promotes epicutaneous antigen sensitization concomitant with the development of food allergy.J Allergy Clin Immunol. 2018 Jan;141(1):223-234.e5. doi: 10.1016/j.jaci.2017.02.035. Epub 2017 Apr 6. J Allergy Clin Immunol. 2018. PMID: 28390860
-
Exposure to food allergens through inflamed skin promotes intestinal food allergy through the thymic stromal lymphopoietin-basophil axis.J Allergy Clin Immunol. 2014 May;133(5):1390-9, 1399.e1-6. doi: 10.1016/j.jaci.2014.01.021. Epub 2014 Feb 20. J Allergy Clin Immunol. 2014. PMID: 24560412 Free PMC article.
Cited by
-
Cholesterol negatively regulates IL-9-producing CD8+ T cell differentiation and antitumor activity.J Exp Med. 2018 Jun 4;215(6):1555-1569. doi: 10.1084/jem.20171576. Epub 2018 May 9. J Exp Med. 2018. PMID: 29743292 Free PMC article.
-
Interleukin-33: Its Emerging Role in Allergic Diseases.Molecules. 2018 Jul 9;23(7):1665. doi: 10.3390/molecules23071665. Molecules. 2018. PMID: 29987222 Free PMC article. Review.
-
IL-9 and Th9 cells in health and diseases-From tolerance to immunopathology.Cytokine Growth Factor Rev. 2017 Oct;37:47-55. doi: 10.1016/j.cytogfr.2017.07.004. Epub 2017 Jul 17. Cytokine Growth Factor Rev. 2017. PMID: 28739029 Free PMC article. Review.
-
The Role of Type 2 Innate Lymphoid Cells in Allergic Diseases.Front Immunol. 2021 Jun 9;12:586078. doi: 10.3389/fimmu.2021.586078. eCollection 2021. Front Immunol. 2021. PMID: 34177881 Free PMC article. Review.
-
Interactions between Innate Lymphoid Cells and Cells of the Innate and Adaptive Immune System.Front Immunol. 2017 Oct 30;8:1422. doi: 10.3389/fimmu.2017.01422. eCollection 2017. Front Immunol. 2017. PMID: 29163497 Free PMC article. Review.
References
-
- Gupta RS, et al. The prevalence, severity, and distribution of childhood food allergy in the United States. Pediatrics. 2011;128(1):e9–17. - PubMed
-
- Sicherer SH, Sampson HA. Food allergy: recent advances in pathophysiology and treatment. Annual review of medicine. 2009;60:261–77. - PubMed
-
- Burbank AJ, et al. Oral Immunotherapy for Food Allergy. Immunol Allergy Clin North Am. 2016;36(1):55–69. - PubMed
-
- Wood RA. Food allergen immunotherapy: Current status and prospects for the future. J Allergy Clin Immunol. 2016;137(4):973–82. - PubMed
-
- Burks AW, Sampson HA. Anaphylaxis and food allergy. Clinical reviews in allergy & immunology. 1999;17(3):339–60. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

