β-Arrestin mediates the Frank-Starling mechanism of cardiac contractility
- PMID: 27911784
- PMCID: PMC5167158
- DOI: 10.1073/pnas.1609308113
β-Arrestin mediates the Frank-Starling mechanism of cardiac contractility
Abstract
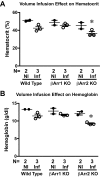
The Frank-Starling law of the heart is a physiological phenomenon that describes an intrinsic property of heart muscle in which increased cardiac filling leads to enhanced cardiac contractility. Identified more than a century ago, the Frank-Starling relationship is currently known to involve length-dependent enhancement of cardiac myofilament Ca2+ sensitivity. However, the upstream molecular events that link cellular stretch to the length-dependent myofilament Ca2+ sensitivity are poorly understood. Because the angiotensin II type 1 receptor (AT1R) and the multifunctional transducer protein β-arrestin have been shown to mediate mechanosensitive cellular signaling, we tested the hypothesis that these two proteins are involved in the Frank-Starling mechanism of the heart. Using invasive hemodynamics, we found that mice lacking β-arrestin 1, β-arrestin 2, or AT1R were unable to generate a Frank-Starling force in response to changes in cardiac volume. Although wild-type mice pretreated with the conventional AT1R blocker losartan were unable to enhance cardiac contractility with volume loading, treatment with a β-arrestin-biased AT1R ligand to selectively activate β-arrestin signaling preserved the Frank-Starling relationship. Importantly, in skinned muscle fiber preparations, we found markedly impaired length-dependent myofilament Ca2+ sensitivity in β-arrestin 1, β-arrestin 2, and AT1R knockout mice. Our data reveal β-arrestin 1, β-arrestin 2, and AT1R as key regulatory molecules in the Frank-Starling mechanism, which potentially can be targeted therapeutically with β-arrestin-biased AT1R ligands.
Keywords: angiotensin II type I receptor; cardiac function; hemodynamics; mechanotransduction; β-arrestin.
Conflict of interest statement
H.A.R. is a scientific cofounder of Trevena, Inc., a company developing G protein-coupled receptor targeted drugs.
Figures
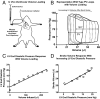
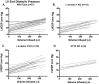
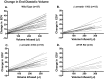
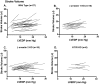






References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

