Chloroquine, an Endocytosis Blocking Agent, Inhibits Zika Virus Infection in Different Cell Models
- PMID: 27916837
- PMCID: PMC5192383
- DOI: 10.3390/v8120322
Chloroquine, an Endocytosis Blocking Agent, Inhibits Zika Virus Infection in Different Cell Models
Abstract
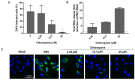
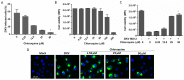
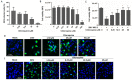
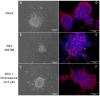
Zika virus (ZIKV) infection in utero might lead to microcephaly and other congenital defects. Since no specific therapy is available thus far, there is an urgent need for the discovery of agents capable of inhibiting its viral replication and deleterious effects. Chloroquine is widely used as an antimalarial drug, anti-inflammatory agent, and it also shows antiviral activity against several viruses. Here we show that chloroquine exhibits antiviral activity against ZIKV in Vero cells, human brain microvascular endothelial cells, human neural stem cells, and mouse neurospheres. We demonstrate that chloroquine reduces the number of ZIKV-infected cells in vitro, and inhibits virus production and cell death promoted by ZIKV infection without cytotoxic effects. In addition, chloroquine treatment partially reveres morphological changes induced by ZIKV infection in mouse neurospheres.
Keywords: Zika virus; antiviral; chloroquine; microcephaly; neural stem cell.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
-
- Lanciotti R.S., Kosoy O.L., Laven J.J., Velez J.O., Lambert A.J., Johnson A.J., Stanfield S.M., Duffy M.R. Genetic and serologic properties of Zika virus associated with an epidemic, Yap State, Micronesia, 2007. Emerg. Infect. Dis. 2008;14:1232–1239. doi: 10.3201/eid1408.080287. - DOI - PMC - PubMed
-
- Brasil P., Pereira J.P., Jr., Raja Gabaglia C., Damasceno L., Wakimoto M., Ribeiro Nogueira R.M., Carvalho de Sequeira P., Machado Siqueira A., Abreu de Carvalho L.M., Cotrim da Cunha D., et al. Zika Virus Infection in Pregnant Women in Rio de Janeiro—Preliminary Report. N. Engl. J. Med. 2016 doi: 10.1056/NEJMoa1602412. - DOI - PMC - PubMed
-
- Martines R.B., Bhatnagar J., Keating M.K., Silva-Flannery L., Muehlenbachs A., Gary J., Goldsmith C., Hale G., Ritter J., Rollin D., et al. Notes from the Field: Evidence of Zika Virus Infection in Brain and Placental Tissues from Two Congenitally Infected Newborns and Two Fetal Losses —Brazil, 2015. MMWR Morb. Mortal. Wkly. Rep. 2016;65:1–2. doi: 10.15585/mmwr.mm6506e1er. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

