Dysregulated Homeostasis of Acetylcholine Levels in Immune Cells of RR-Multiple Sclerosis Patients
- PMID: 27916909
- PMCID: PMC5187809
- DOI: 10.3390/ijms17122009
Dysregulated Homeostasis of Acetylcholine Levels in Immune Cells of RR-Multiple Sclerosis Patients
Abstract
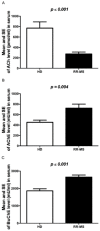
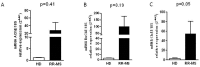
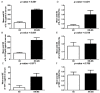
Multiple sclerosis (MS) is characterized by pro-inflammatory cytokine production. Acetylcholine (ACh) contributes to the modulation of central and peripheral inflammation. We studied the homeostasis of the cholinergic system in relation to cytokine levels in immune cells and sera of relapsing remitting-MS (RR-MS) patients. We demonstrated that lower ACh levels in serum of RR-MS patients were inversely correlated with the increased activity of the hydrolyzing enzymes acetylcholinesterase (AChE) and butyrylcholinesterase (BuChE). Interestingly, the expression of the ACh biosynthetic enzyme and the protein carriers involved in non-vesicular ACh release were found overexpressed in peripheral blood mononuclear cells of MS patients. The inflammatory state of the MS patients was confirmed by increased levels of TNFα, IL-12/IL-23p40, IL-18. The lower circulating ACh levels in sera of MS patients are dependent on the higher activity of cholinergic hydrolyzing enzymes. The smaller ratio of ACh to TNFα, IL-12/IL-23p40 and IL-18 in MS patients, with respect to healthy donors (HD), is indicative of an inflammatory environment probably related to the alteration of cholinergic system homeostasis.
Keywords: acetylcholine; acetylcholinesterase; butyrylcholinesterase; cytokines; multiple sclerosis.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Profiling of Canonical and Non-Traditional Cytokine Levels in Interferon-β-Treated Relapsing-Remitting-Multiple Sclerosis Patients.Front Immunol. 2018 Jun 4;9:1240. doi: 10.3389/fimmu.2018.01240. eCollection 2018. Front Immunol. 2018. PMID: 29915590 Free PMC article.
-
Nicotinic receptor activation negatively modulates pro-inflammatory cytokine production in multiple sclerosis patients.Int Immunopharmacol. 2015 Nov;29(1):152-7. doi: 10.1016/j.intimp.2015.06.034. Epub 2015 Jul 23. Int Immunopharmacol. 2015. PMID: 26209886
-
Relation between pro-inflammatory cytokines and acetylcholine levels in relapsing-remitting multiple sclerosis patients.Int J Mol Sci. 2012 Oct 3;13(10):12656-64. doi: 10.3390/ijms131012656. Int J Mol Sci. 2012. PMID: 23202919 Free PMC article.
-
Acetylcholinesterase and butyrylcholinesterase as possible markers of low-grade systemic inflammation.Med Sci Monit. 2007 Dec;13(12):RA214-21. Med Sci Monit. 2007. PMID: 18049445 Review.
-
Possible Correlation between Cholinergic System Alterations and Neuro/Inflammation in Multiple Sclerosis.Biomedicines. 2020 Jun 8;8(6):153. doi: 10.3390/biomedicines8060153. Biomedicines. 2020. PMID: 32521719 Free PMC article. Review.
Cited by
-
Neuroinflammation Modulation via α7 Nicotinic Acetylcholine Receptor and Its Chaperone, RIC-3.Molecules. 2021 Oct 11;26(20):6139. doi: 10.3390/molecules26206139. Molecules. 2021. PMID: 34684720 Free PMC article. Review.
-
Butyrylcholinesterase and Acetylcholinesterase polymorphisms in Multiple Sclerosis patients: implication in peripheral inflammation.Sci Rep. 2018 Jan 22;8(1):1319. doi: 10.1038/s41598-018-19701-7. Sci Rep. 2018. PMID: 29358722 Free PMC article.
-
Role of the Cholinergic Anti-Inflammatory Reflex in Central Nervous System Diseases.Int J Mol Sci. 2021 Dec 14;22(24):13427. doi: 10.3390/ijms222413427. Int J Mol Sci. 2021. PMID: 34948222 Free PMC article. Review.
-
Targeting the gut to treat multiple sclerosis.J Clin Invest. 2021 Jul 1;131(13):e143774. doi: 10.1172/JCI143774. J Clin Invest. 2021. PMID: 34196310 Free PMC article. Review.
-
Acetylcholinesterase Inhibitors in the Treatment of Neurodegenerative Diseases and the Role of Acetylcholinesterase in their Pathogenesis.Int J Mol Sci. 2021 Aug 27;22(17):9290. doi: 10.3390/ijms22179290. Int J Mol Sci. 2021. PMID: 34502198 Free PMC article. Review.
References
-
- Martins T.B., Rose J.W., Jaskowski T.D., Wilson A.R., Husebye D., Seraj H.S., Hill H.R. Analysis of proinflammatory and anti-inflammatory cytokine serum concentrations in patients with multiple sclerosis by using a multiplexed immunoassay. Am. J. Clin. Pathol. 2011;136:696–704. doi: 10.1309/AJCP7UBK8IBVMVNR. - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

