EEG Negativity in Fixations Used for Gaze-Based Control: Toward Converting Intentions into Actions with an Eye-Brain-Computer Interface
- PMID: 27917105
- PMCID: PMC5114310
- DOI: 10.3389/fnins.2016.00528
EEG Negativity in Fixations Used for Gaze-Based Control: Toward Converting Intentions into Actions with an Eye-Brain-Computer Interface
Abstract
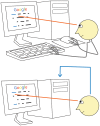
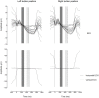
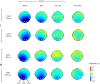
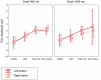
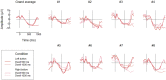
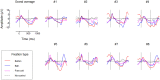
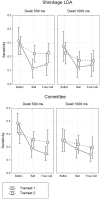
We usually look at an object when we are going to manipulate it. Thus, eye tracking can be used to communicate intended actions. An effective human-machine interface, however, should be able to differentiate intentional and spontaneous eye movements. We report an electroencephalogram (EEG) marker that differentiates gaze fixations used for control from spontaneous fixations involved in visual exploration. Eight healthy participants played a game with their eye movements only. Their gaze-synchronized EEG data (fixation-related potentials, FRPs) were collected during game's control-on and control-off conditions. A slow negative wave with a maximum in the parietooccipital region was present in each participant's averaged FRPs in the control-on conditions and was absent or had much lower amplitude in the control-off condition. This wave was similar but not identical to stimulus-preceding negativity, a slow negative wave that can be observed during feedback expectation. Classification of intentional vs. spontaneous fixations was based on amplitude features from 13 EEG channels using 300 ms length segments free from electrooculogram contamination (200-500 ms relative to the fixation onset). For the first fixations in the fixation triplets required to make moves in the game, classified against control-off data, a committee of greedy classifiers provided 0.90 ± 0.07 specificity and 0.38 ± 0.14 sensitivity. Similar (slightly lower) results were obtained for the shrinkage Linear Discriminate Analysis (LDA) classifier. The second and third fixations in the triplets were classified at lower rate. We expect that, with improved feature sets and classifiers, a hybrid dwell-based Eye-Brain-Computer Interface (EBCI) can be built using the FRP difference between the intended and spontaneous fixations. If this direction of BCI development will be successful, such a multimodal interface may improve the fluency of interaction and can possibly become the basis for a new input device for paralyzed and healthy users, the EBCI "Wish Mouse."
Keywords: assistive technology; brain-computer interfaces; detection of intention; eye tracking; gaze interaction; human-computer interfaces; slow cortical potentials; stimulus-preceding negativity.
Figures

Similar articles
-
MEG-Based Detection of Voluntary Eye Fixations Used to Control a Computer.Front Neurosci. 2021 Feb 5;15:619591. doi: 10.3389/fnins.2021.619591. eCollection 2021. Front Neurosci. 2021. PMID: 33613182 Free PMC article.
-
A passive BCI for monitoring the intentionality of the gaze-based moving object selection.J Neural Eng. 2021 Mar 4;18(2). doi: 10.1088/1741-2552/abda09. J Neural Eng. 2021. PMID: 33418554
-
Toward FRP-Based Brain-Machine Interfaces-Single-Trial Classification of Fixation-Related Potentials.PLoS One. 2016 Jan 26;11(1):e0146848. doi: 10.1371/journal.pone.0146848. eCollection 2016. PLoS One. 2016. PMID: 26812487 Free PMC article.
-
Coregistration of eye movements and EEG in natural reading: analyses and review.J Exp Psychol Gen. 2011 Nov;140(4):552-72. doi: 10.1037/a0023885. J Exp Psychol Gen. 2011. PMID: 21744985 Review.
-
Eye-Tracking Feature Extraction for Biometric Machine Learning.Front Neurorobot. 2022 Feb 1;15:796895. doi: 10.3389/fnbot.2021.796895. eCollection 2021. Front Neurorobot. 2022. PMID: 35177973 Free PMC article. Review.
Cited by
-
Hybrid Brain-Computer Interface Techniques for Improved Classification Accuracy and Increased Number of Commands: A Review.Front Neurorobot. 2017 Jul 24;11:35. doi: 10.3389/fnbot.2017.00035. eCollection 2017. Front Neurorobot. 2017. PMID: 28790910 Free PMC article. Review.
-
Using Fixation-Related Potentials for Inspecting Natural Interactions.Front Hum Neurosci. 2020 Nov 5;14:579505. doi: 10.3389/fnhum.2020.579505. eCollection 2020. Front Hum Neurosci. 2020. PMID: 33250729 Free PMC article.
-
MEG-Based Detection of Voluntary Eye Fixations Used to Control a Computer.Front Neurosci. 2021 Feb 5;15:619591. doi: 10.3389/fnins.2021.619591. eCollection 2021. Front Neurosci. 2021. PMID: 33613182 Free PMC article.
-
Active Brain-Computer Interfacing for Healthy Users.Front Neurosci. 2022 Apr 25;16:859887. doi: 10.3389/fnins.2022.859887. eCollection 2022. Front Neurosci. 2022. PMID: 35546879 Free PMC article. No abstract available.
-
A P300 Brain-Computer Interface With a Reduced Visual Field.Front Neurosci. 2020 Dec 3;14:604629. doi: 10.3389/fnins.2020.604629. eCollection 2020. Front Neurosci. 2020. PMID: 33343290 Free PMC article.
References
-
- Banville H., Falk T. H. (2016). Recent advances and open challenges in hybrid brain-computer interfacing: a technological review of non-invasive human research. Brain Comput. Interfaces 3, 9–46. 10.1080/2326263X.2015.1134958 - DOI
-
- Blankertz B., Curio G., Muller K. R. (2002). Classifying single trial EEG: Towards brain computer interfacing. Adv. Neural Inf. Process. Syst. 14, 157–164.
-
- Blankertz B., Dornhege G., Schäfer C., Krepki R., Kohlmorgen J., Müller K. R., et al. . (2003). Boosting bit rates and error detection for the classification of fast-paced motor commands based on single-trial EEG analysis. IEEE Trans. Neural Syst. Rehabil. Eng. 11, 127–131. 10.1109/TNSRE.2003.814456 - DOI - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

