Three-dimensional Modelling of the Voltage-gated Sodium Ion Channel from Anopheles gambiae Reveals Spatial Clustering of Evolutionarily Conserved Acidic Residues at the Extracellular Sites
- PMID: 27919210
- PMCID: PMC5725538
- DOI: 10.2174/1567201814666161205131213
Three-dimensional Modelling of the Voltage-gated Sodium Ion Channel from Anopheles gambiae Reveals Spatial Clustering of Evolutionarily Conserved Acidic Residues at the Extracellular Sites
Abstract
Background: The eukaryotic voltage-gated sodium channel(e-Nav) is a large asymmetric transmembrane protein with important functions concerning neurological function. No structure has been resolved at high resolution for this protein.
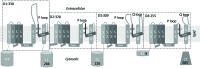
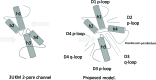
Methods: A homology model of the transmembrane and extracellular regions of an Anopheles gambiae para-like channel with emphasis on the pore entrance has been constructed, based upon the templates provided by a prokaryotic sodium channel and a potassium two-pore channel. The latter provides a template for the extracellular regions, which are located above the entrance to the pore, which is likely to open at a side of a dome formed by these loops.
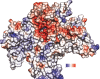
Results: A model created with this arrangement shows a structure similar to low-resolution cryoelectron microscope images of a related structure. The pore entrance also shows favorable electrostatic interface.
Conclusion: Residues responsible for the negative charge around the pore have been traced in phylogeny to highlight their importance. This model is intended for the study of pore-blocking toxins.
Keywords: Eukaryotic voltage gated sodium channel; anopheles.; extracellular interface; homology model; pore blocker; toxin; transmembrane.
Copyright© Bentham Science Publishers; For any queries, please email at epub@benthamscience.org.
Figures





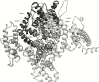
Similar articles
-
Brownian dynamics simulations of the recognition of the scorpion toxin maurotoxin with the voltage-gated potassium ion channels.Biophys J. 2002 Nov;83(5):2370-85. doi: 10.1016/S0006-3495(02)75251-X. Biophys J. 2002. PMID: 12414674 Free PMC article.
-
Emerging issues of connexin channels: biophysics fills the gap.Q Rev Biophys. 2001 Aug;34(3):325-472. doi: 10.1017/s0033583501003705. Q Rev Biophys. 2001. PMID: 11838236 Review.
-
Molecular pore structure of voltage-gated sodium and calcium channels.Braz J Med Biol Res. 1994 Dec;27(12):2781-802. Braz J Med Biol Res. 1994. PMID: 7550000 Review.
-
Mapping of scorpion toxin receptor sites at voltage-gated sodium channels.Toxicon. 2012 Sep 15;60(4):502-11. doi: 10.1016/j.toxicon.2012.03.022. Epub 2012 Apr 4. Toxicon. 2012. PMID: 22694883
-
Solution structure of the sodium channel antagonist conotoxin GS: a new molecular caliper for probing sodium channel geometry.Structure. 1997 Apr 15;5(4):571-83. doi: 10.1016/s0969-2126(97)00212-8. Structure. 1997. PMID: 9115446
Cited by
-
Editorial: Improving Neuropharmacology using Big Data, Machine Learning and Computational Algorithms.Curr Neuropharmacol. 2017 Nov 14;15(8):1058-1061. doi: 10.2174/1570159X1508171114113425. Curr Neuropharmacol. 2017. PMID: 29199918 Free PMC article. No abstract available.
References
-
- Hodgkin A.L., Huxley A.F., Katz B. Measurement of current-voltage relations in the membrane of the giant axon of Loligo. J. Physiol. 1952;116(4):424–448. [http://dx.doi.org/10.1113/ jphysiol.1952.sp004716]. [PMID: 14946712]. - PMC - PubMed
-
- Hodgkin A.L., Huxley A.F. Currents carried by sodium and potassium ions through the membrane of the giant axon of Loligo. J. Physiol. 1952;116(4):449–472. [http://dx.doi.org/10.1113/ jphysiol.1952.sp004717]. [PMID: 14946713]. - PMC - PubMed
-
- Hodgkin A.L., Huxley A.F. The dual effect of membrane potential on sodium conductance in the giant axon of Loligo. J. Physiol. 1952;116(4):497–506. [http://dx.doi.org/10.1113/jphysiol. 1952.sp004719]. [PMID: 14946715]. - PMC - PubMed
-
- Duclohier H. Structure-function studies on the voltage-gated sodium channel. Biochim. Biophys. Acta. 2009;1788(11):2374–2379. [http://dx.doi.org/10.1016/j.bbamem.2009.08.017]. [PMID: 19747894]. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
