In vitro activation of coagulation by human neutrophil DNA and histone proteins but not neutrophil extracellular traps
- PMID: 27919911
- PMCID: PMC5324715
- DOI: 10.1182/blood-2016-06-722298
In vitro activation of coagulation by human neutrophil DNA and histone proteins but not neutrophil extracellular traps
Abstract
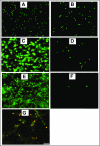
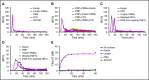
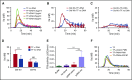
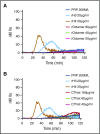
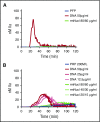
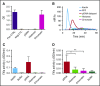
NETosis is a physiologic process in which neutrophils release their nuclear material in the form of neutrophil extracellular traps (NETs). NETs have been reported to directly promote thrombosis in animal models. Although the effects of purified NET components including DNA, histone proteins, and neutrophil enzymes on coagulation have been characterized, the mechanism by which intact NETs promote thrombosis is largely unknown. In this study, human neutrophils were stimulated to produce NETs in platelet-free plasma (PFP) or in buffer using phorbol myristate actetate or calcium ionophore. DNA and histone proteins were also separately purified from normal human neutrophils and used to reconstitute chromatin using a salt-gradient dialysis method. Neutrophil stimulation resulted in robust NET release. In recalcified PFP, purified DNA triggered contact-dependent thrombin generation (TG) and amplified TG initiated by low concentrations of tissue factor. Similarly, in a buffer milieu, DNA initiated the contact pathway and amplified thrombin-dependent factor XI activation. Recombinant human histones H3 and H4 triggered TG in recalcified human plasma in a platelet-dependent manner. In contrast, neither intact NETs, reconstituted chromatin, individual nucleosome particles, nor octameric core histones reproduced any of these procoagulant effects. We conclude that unlike DNA or individual histone proteins, human intact NETs do not directly initiate or amplify coagulation in vitro. This difference is likely explained by the complex histone-histone and histone-DNA interactions within the nucleosome unit and higher-order supercoiled chromatin leading to neutralization of the negative charges on polyanionic DNA and modification of the binding properties of individual histone proteins.
© 2017 by The American Society of Hematology.
Figures

Comment in
-
Demystifying the prothrombotic role of NETs.Blood. 2017 Feb 23;129(8):925-926. doi: 10.1182/blood-2017-01-757328. Blood. 2017. PMID: 28232623 No abstract available.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

