Exercise-induced stress behavior, gut-microbiota-brain axis and diet: a systematic review for athletes
- PMID: 27924137
- PMCID: PMC5121944
- DOI: 10.1186/s12970-016-0155-6
Exercise-induced stress behavior, gut-microbiota-brain axis and diet: a systematic review for athletes
Abstract
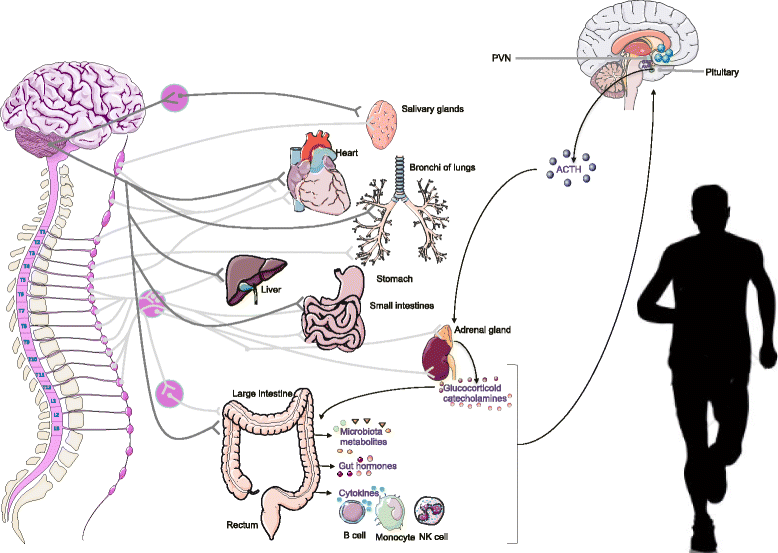
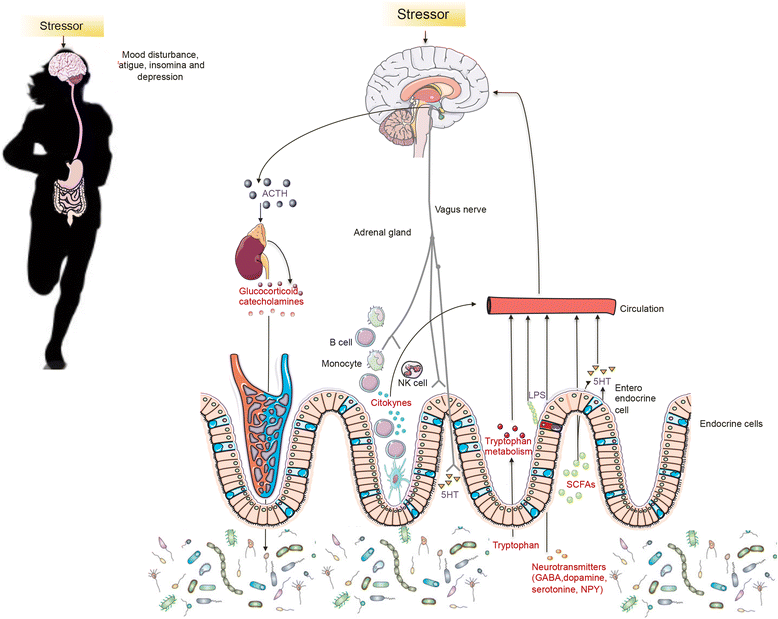
Fatigue, mood disturbances, under performance and gastrointestinal distress are common among athletes during training and competition. The psychosocial and physical demands during intense exercise can initiate a stress response activating the sympathetic-adrenomedullary and hypothalamus-pituitary-adrenal (HPA) axes, resulting in the release of stress and catabolic hormones, inflammatory cytokines and microbial molecules. The gut is home to trillions of microorganisms that have fundamental roles in many aspects of human biology, including metabolism, endocrine, neuronal and immune function. The gut microbiome and its influence on host behavior, intestinal barrier and immune function are believed to be a critical aspect of the brain-gut axis. Recent evidence in murine models shows that there is a high correlation between physical and emotional stress during exercise and changes in gastrointestinal microbiota composition. For instance, induced exercise-stress decreased cecal levels of Turicibacter spp and increased Ruminococcus gnavus, which have well defined roles in intestinal mucus degradation and immune function. Diet is known to dramatically modulate the composition of the gut microbiota. Due to the considerable complexity of stress responses in elite athletes (from leaky gut to increased catabolism and depression), defining standard diet regimes is difficult. However, some preliminary experimental data obtained from studies using probiotics and prebiotics studies show some interesting results, indicating that the microbiota acts like an endocrine organ (e.g. secreting serotonin, dopamine or other neurotransmitters) and may control the HPA axis in athletes. What is troubling is that dietary recommendations for elite athletes are primarily based on a low consumption of plant polysaccharides, which is associated with reduced microbiota diversity and functionality (e.g. less synthesis of byproducts such as short chain fatty acids and neurotransmitters). As more elite athletes suffer from psychological and gastrointestinal conditions that can be linked to the gut, targeting the microbiota therapeutically may need to be incorporated in athletes' diets that take into consideration dietary fiber as well as microbial taxa not currently present in athlete's gut.
Keywords: Athlete; Behaviour; Diet; Exercise; Microbiota; Neurotransmitters; Stress.
Figures

References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous
