A LON-ClpP Proteolytic Axis Degrades Complex I to Extinguish ROS Production in Depolarized Mitochondria
- PMID: 27926857
- PMCID: PMC5177631
- DOI: 10.1016/j.celrep.2016.11.027
A LON-ClpP Proteolytic Axis Degrades Complex I to Extinguish ROS Production in Depolarized Mitochondria
Abstract
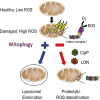
Mitochondrial dysfunction is implicated in numerous neurodegenerative disorders and in Parkinson's disease (PD) in particular. PINK1 and Parkin gene mutations are causes of autosomal recessive PD, and these respective proteins function cooperatively to degrade depolarized mitochondria (mitophagy). It is widely assumed that impaired mitophagy causes PD, as toxic reactive oxygen species (ROS)-producing mitochondria accumulate and progressively drive neurodegeneration. Instead, we report that a LON-ClpP proteolytic quality control axis extinguishes ROS in depolarized mitochondria by degrading the complex I ROS-generating domain. Complex I deficiency has also been identified in PD brain, and our study provides a compelling non-genetic mechanistic rationale to explain this observation: intact complex I depletes if mitochondrial bioenergetic capacity is robustly attenuated.
Keywords: ClpP; LON; NADH:ubiquinone oxidoreductase; complex I; mitochondria; mitochondrial proteases; mitophagy; retrograde signaling.
Copyright © 2016 The Author(s). Published by Elsevier Inc. All rights reserved.
Figures


Similar articles
-
The complex I subunit NDUFA10 selectively rescues Drosophila pink1 mutants through a mechanism independent of mitophagy.PLoS Genet. 2014 Nov 20;10(11):e1004815. doi: 10.1371/journal.pgen.1004815. eCollection 2014 Nov. PLoS Genet. 2014. PMID: 25412178 Free PMC article.
-
Nix restores mitophagy and mitochondrial function to protect against PINK1/Parkin-related Parkinson's disease.Sci Rep. 2017 Mar 10;7:44373. doi: 10.1038/srep44373. Sci Rep. 2017. PMID: 28281653 Free PMC article.
-
The PINK1/Parkin-mediated mitophagy is compromised by PD-associated mutations.Autophagy. 2010 Oct;6(7):871-8. doi: 10.4161/auto.6.7.13286. Epub 2010 Oct 3. Autophagy. 2010. PMID: 20798600
-
N-degron-mediated degradation and regulation of mitochondrial PINK1 kinase.Curr Genet. 2020 Aug;66(4):693-701. doi: 10.1007/s00294-020-01062-2. Epub 2020 Mar 10. Curr Genet. 2020. PMID: 32157382 Review.
-
Parkin and PINK1 functions in oxidative stress and neurodegeneration.Brain Res Bull. 2017 Jul;133:51-59. doi: 10.1016/j.brainresbull.2016.12.004. Epub 2016 Dec 23. Brain Res Bull. 2017. PMID: 28017782 Free PMC article. Review.
Cited by
-
Quality control of the mitochondrial proteome.Nat Rev Mol Cell Biol. 2021 Jan;22(1):54-70. doi: 10.1038/s41580-020-00300-2. Epub 2020 Oct 22. Nat Rev Mol Cell Biol. 2021. PMID: 33093673 Review.
-
Common gene expression signatures in Parkinson's disease are driven by changes in cell composition.Acta Neuropathol Commun. 2020 Apr 21;8(1):55. doi: 10.1186/s40478-020-00932-7. Acta Neuropathol Commun. 2020. PMID: 32317022 Free PMC article.
-
OMA1 protease eliminates arrested protein import intermediates upon mitochondrial depolarization.J Cell Biol. 2024 May 6;223(5):e202306051. doi: 10.1083/jcb.202306051. Epub 2024 Mar 26. J Cell Biol. 2024. PMID: 38530280 Free PMC article.
-
AAA+ proteases: the first line of defense against mitochondrial damage.PeerJ. 2022 Nov 7;10:e14350. doi: 10.7717/peerj.14350. eCollection 2022. PeerJ. 2022. PMID: 36389399 Free PMC article. Review.
-
Mitochondrial Calcium Regulation of Cardiac Metabolism in Health and Disease.Physiology (Bethesda). 2024 Sep 1;39(5):0. doi: 10.1152/physiol.00014.2024. Epub 2024 May 7. Physiology (Bethesda). 2024. PMID: 38713090 Review.
References
-
- Bota D.A., Davies K.J. Lon protease preferentially degrades oxidized mitochondrial aconitase by an ATP-stimulated mechanism. Nat. Cell Biol. 2002;4:674–680. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

