Analysis of 138 pathogenic mutations in presenilin-1 on the in vitro production of Aβ42 and Aβ40 peptides by γ-secretase
- PMID: 27930341
- PMCID: PMC5278480
- DOI: 10.1073/pnas.1618657114
Analysis of 138 pathogenic mutations in presenilin-1 on the in vitro production of Aβ42 and Aβ40 peptides by γ-secretase
Abstract
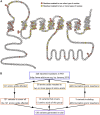
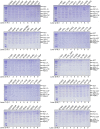
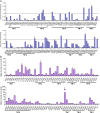
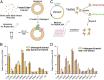
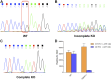
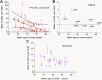
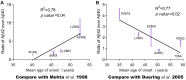
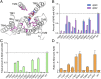
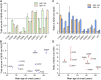
A hallmark of Alzheimer's disease (AD) is the aggregation of β-amyloid peptides (Aβ) into amyloid plaques in patient brain. Cleavage of amyloid precursor protein (APP) by the intramembrane protease γ-secretase produces Aβ of varying lengths, of which longer peptides such as Aβ42 are thought to be more harmful. Increased ratios of longer Aβs over shorter ones, exemplified by the ratio of Aβ42 over Aβ40, may lead to formation of amyloid plaques and consequent development of AD. In this study, we analyzed 138 reported mutations in human presenilin-1 (PS1) by individually reconstituting the mutant PS1 proteins into anterior-pharynx-defective protein 1 (APH-1)aL-containing γ-secretases and examining their abilities to produce Aβ42 and Aβ40 in vitro. About 90% of these mutations lead to reduced production of Aβ42 and Aβ40. Notably, 10% of these mutations result in decreased Aβ42/Aβ40 ratios. There is no statistically significant correlation between the Aβ42/Aβ40 ratio produced by a γ-secretase variant containing a specific PS1 mutation and the mean age at onset of patients from whom the mutation was isolated.
Keywords: Alzheimer’s disease; Aβ peptides; amyloid hypothesis; cleavage activity; γ-secretase.
Conflict of interest statement
The authors declare no conflict of interest.
Figures






Comment in
-
Presenilin-1 mutations and Alzheimer's disease.Proc Natl Acad Sci U S A. 2017 Jan 24;114(4):629-631. doi: 10.1073/pnas.1619574114. Epub 2017 Jan 12. Proc Natl Acad Sci U S A. 2017. PMID: 28082723 Free PMC article. No abstract available.
-
Aβ42/Aβ40 Ratios of Presenilin 1 Mutations Correlate with Clinical Onset of Alzheimer's Disease.J Alzheimers Dis. 2018;66(3):939-945. doi: 10.3233/JAD-180829. J Alzheimers Dis. 2018. PMID: 30412504
References
-
- Alzheimer A. About a peculiar disease of the cerebral cortex. Centralblatt für Nervenheilkunde Psychiatrie. 1907;30:177–179.
-
- Glenner GG, Wong CW. Alzheimer’s disease and Down’s syndrome: Sharing of a unique cerebrovascular amyloid fibril protein. Biochem Biophys Res Commun. 1984;122(3):1131–1135. - PubMed
-
- Estus S, et al. Potentially amyloidogenic, carboxyl-terminal derivatives of the amyloid protein precursor. Science. 1992;255(5045):726–728. - PubMed
-
- Golde TE, Estus S, Younkin LH, Selkoe DJ, Younkin SG. Processing of the amyloid protein precursor to potentially amyloidogenic derivatives. Science. 1992;255(5045):728–730. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

