Local Vascular Gene Therapy With Apolipoprotein A-I to Promote Regression of Atherosclerosis
- PMID: 27932352
- PMCID: PMC5269454
- DOI: 10.1161/ATVBAHA.116.308258
Local Vascular Gene Therapy With Apolipoprotein A-I to Promote Regression of Atherosclerosis
Abstract
Objective: Gene therapy, delivered directly to the blood vessel wall, could potentially prevent atherosclerotic lesion growth and promote atherosclerosis regression. Previously, we reported that a helper-dependent adenoviral (HDAd) vector expressing apolipoprotein A-I (apoA-I) in carotid endothelium of fat-fed rabbits reduced early (4 weeks) atherosclerotic lesion growth. Here, we tested whether the same HDAd-delivered to the existing carotid atherosclerotic lesions-could promote regression.
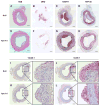
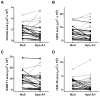
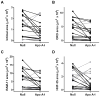
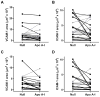
Approach and results: Rabbits (n=26) were fed a high-fat diet for 7 months, then treated with bilateral carotid gene transfer. One carotid was infused with an HDAd expressing apoA-I (HDAdApoAI) and the other with a control nonexpressing HDAd (HDAdNull). The side with HDAdApoAI was randomized. Rabbits were then switched to regular chow, lowering their plasma cholesterols by over 70%. ApoA-I mRNA and protein were detected in HDAdApoAI-transduced arteries. After 7 weeks of gene therapy, compared with HDAdNull-treated arteries in the same rabbits, HDAdApoAI-treated arteries had significantly less vascular cell adhesion molecule-1 expression (28%; P=0.04) along with modest but statistically insignificant trends toward decreased intimal lesion volume, lipid and macrophage content, and intercellular adhesion molecule-1 expression (9%-21%; P=0.1-0.4). Post hoc subgroup analysis of rabbits with small-to-moderate-sized lesions (n=20) showed that HDAdApoAI caused large reductions in lesion volume, lipid content, intercellular adhesion molecule-1, and vascular cell adhesion molecule-1 expression (30%-50%; P≤0.04 for all). Macrophage content was reduced by 30% (P=0.06). There was a significant interaction (P=0.02) between lesion size and treatment efficacy.
Conclusions: Even when administered on a background of aggressive lowering of plasma cholesterol, local HDAdApoAI vascular gene therapy may promote rapid regression of small-to-moderate-sized atherosclerotic lesions.
Keywords: apolipoprotein A-I; atherosclerosis; carotid arteries; genetic therapy; macrophages; rabbits.
© 2016 American Heart Association, Inc.
Conflict of interest statement
The authors have no conflicts to disclose.
Figures


Similar articles
-
Apo A-I (Apolipoprotein A-I) Vascular Gene Therapy Provides Durable Protection Against Atherosclerosis in Hyperlipidemic Rabbits.Arterioscler Thromb Vasc Biol. 2018 Jan;38(1):206-217. doi: 10.1161/ATVBAHA.117.309565. Epub 2017 Nov 9. Arterioscler Thromb Vasc Biol. 2018. PMID: 29122817 Free PMC article.
-
Expression of apolipoprotein A-I in rabbit carotid endothelium protects against atherosclerosis.Mol Ther. 2011 Oct;19(10):1833-41. doi: 10.1038/mt.2011.133. Epub 2011 Jul 19. Mol Ther. 2011. PMID: 21772254 Free PMC article.
-
NLRP3 Inflammasome Inhibition by MCC950 Reduces Atherosclerotic Lesion Development in Apolipoprotein E-Deficient Mice-Brief Report.Arterioscler Thromb Vasc Biol. 2017 Aug;37(8):1457-1461. doi: 10.1161/ATVBAHA.117.309575. Epub 2017 Jun 8. Arterioscler Thromb Vasc Biol. 2017. PMID: 28596375
-
Regression of Atherosclerosis: The Journey From the Liver to the Plaque and Back.Arterioscler Thromb Vasc Biol. 2016 Feb;36(2):226-35. doi: 10.1161/ATVBAHA.115.301926. Epub 2015 Dec 17. Arterioscler Thromb Vasc Biol. 2016. PMID: 26681754 Free PMC article. Review.
-
MicroRNA Regulation of Atherosclerosis.Circ Res. 2016 Feb 19;118(4):703-20. doi: 10.1161/CIRCRESAHA.115.306300. Circ Res. 2016. PMID: 26892968 Free PMC article. Review.
Cited by
-
Exosome-Mediated Transfer of Anti-miR-33a-5p from Transduced Endothelial Cells Enhances Macrophage and Vascular Smooth Muscle Cell Cholesterol Efflux.Hum Gene Ther. 2020 Feb;31(3-4):219-232. doi: 10.1089/hum.2019.245. Epub 2020 Jan 16. Hum Gene Ther. 2020. PMID: 31842627 Free PMC article.
-
Cardiovascular Gene Therapy: Past, Present, and Future.Mol Ther. 2017 May 3;25(5):1095-1106. doi: 10.1016/j.ymthe.2017.03.027. Epub 2017 Apr 4. Mol Ther. 2017. PMID: 28389321 Free PMC article. Review.
-
Alginate hydrogel polymers enable efficient delivery of a vascular-targeted AAV vector into aortic tissue.Mol Ther Methods Clin Dev. 2021 Feb 24;21:83-93. doi: 10.1016/j.omtm.2021.02.017. eCollection 2021 Jun 11. Mol Ther Methods Clin Dev. 2021. PMID: 33768132 Free PMC article.
-
ABCA1, ABCG1, and Cholesterol Homeostasis.Adv Exp Med Biol. 2022;1377:95-107. doi: 10.1007/978-981-19-1592-5_7. Adv Exp Med Biol. 2022. PMID: 35575923
-
Advances in Atherosclerosis Theranostics Harnessing Iron Oxide-Based Nanoparticles.Adv Sci (Weinh). 2024 May;11(17):e2308298. doi: 10.1002/advs.202308298. Epub 2024 Feb 17. Adv Sci (Weinh). 2024. PMID: 38368274 Free PMC article. Review.
References
-
- Nissen SE, Stroes E, Dent-Acosta RE, et al. Efficacy and tolerability of evolocumab vs ezetimibe in patients with muscle-related statin intolerance: The gauss-3 randomized clinical trial. JAMA. 2016;315:1580–1590. - PubMed
-
- Sible AM, Nawarskas JJ, Anderson JR. Pcsk9 inhibitors: An innovative approach to treating hyperlipidemia. Cardiol Rev. 2016;24:141–152. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

