Early kidney damage induced by subchronic exposure to PM2.5 in rats
- PMID: 27955691
- PMCID: PMC5154051
- DOI: 10.1186/s12989-016-0179-8
Early kidney damage induced by subchronic exposure to PM2.5 in rats
Abstract
Background: Particulate matter exposure is associated with respiratory and cardiovascular system dysfunction. Recently, we demonstrated that fine particles, also named PM2.5, modify the expression of some components of the angiotensin and bradykinin systems, which are involved in lung, cardiac and renal regulation. The endocrine kidney function is associated with the regulation of angiotensin and bradykinin, and it can suffer damage even as a consequence of minor alterations of these systems. We hypothesized that exposure to PM2.5 can contribute to early kidney damage as a consequence of an angiotensin/bradykinin system imbalance, oxidative stress and/or inflammation.
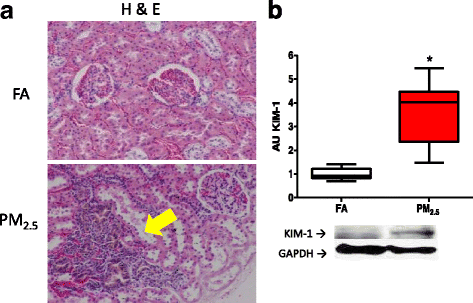
Results: After acute and subchronic exposure to PM2.5, lung damage was confirmed by increased bronchoalveolar lavage fluid (BALF) differential cell counts and a decrease of surfactant protein-A levels. We observed a statistically significant increment in median blood pressure, urine volume and water consumption after PM2.5 exposure. Moreover, increases in the levels of early kidney damage markers were observed after subchronic PM2.5 exposure: the most sensitive markers, β-2-microglobulin and cystatin-C, increased during the first, second, sixth and eighth weeks of exposure. In addition, a reduction in the levels of specific cytokines (IL-1β, IL-6, TNF-α, IL-4, IL-10, INF-γ, IL-17a, MIP-2 and RANTES), and up-regulated angiotensin and bradykinin system markers and indicators of a depleted antioxidant response, were also observed. All of these effects are in concurrence with the presence of renal histological lesions and an early pro-fibrotic state.
Conclusion: Subchronic exposure to PM2.5 induced an early kidney damage response that involved the angiotensin/bradykinin systems as well as antioxidant and immune imbalance. Our study demonstrates that PM2.5 can induce a systemic imbalance that not only affects the cardiovascular system, but also affects the kidney, which may also overall contribute to PM-related diseases.
Keywords: Angiotensin and bradykinin systems; Antioxidant response; Cardiovascular diseases; Inflammation; Kidney biomarkers.
Figures







References
-
- Osornio-Vargas AR, Bonner JC, Alfaro-Moreno E, Martínez L, García-Cuellar C, Rosales SP-d-L, Miranda J, Rosas I. Proinflammatory and cytotoxic effects of Mexico City air pollution particulate matter in vitro are dependent on particle size and composition. Environ Health Perspect. 2003;111:1289. - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

