A Multistrain Mathematical Model To Investigate the Role of Pyrazinamide in the Emergence of Extensively Drug-Resistant Tuberculosis
- PMID: 27956422
- PMCID: PMC5328532
- DOI: 10.1128/AAC.00498-16
A Multistrain Mathematical Model To Investigate the Role of Pyrazinamide in the Emergence of Extensively Drug-Resistant Tuberculosis
Abstract
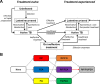
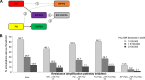
Several infectious diseases of global importance-e.g., HIV infection and tuberculosis (TB)-require prolonged treatment with combination antimicrobial regimens typically involving high-potency core agents coupled with additional companion drugs that protect against the de novo emergence of mutations conferring resistance to the core agents. Often, the most effective (or least toxic) companion agents are reused in sequential (first-line, second-line, etc.) regimens. We used a multistrain model of Mycobacterium tuberculosis transmission in Southeast Asia to investigate how this practice might facilitate the emergence of extensive drug resistance, i.e., resistance to multiple core agents. We calibrated this model to regional TB and drug resistance data using an approximate Bayesian computational approach. We report the proportion of data-consistent simulations in which the prevalence of pre-extensively drug-resistant (pre-XDR) TB-defined as resistance to both first-line and second-line core agents (rifampin and fluoroquinolones)-exceeds predefined acceptability thresholds (1 to 2 cases per 100,000 population by 2035). The use of pyrazinamide (the most effective companion agent) in both first-line and second-line regimens increased the proportion of simulations in which the prevalence exceeded the pre-XDR acceptability threshold by 7-fold compared to a scenario in which patients with pyrazinamide-resistant TB received an alternative drug. Model parameters related to the emergence and transmission of pyrazinamide-resistant TB and resistance amplification were among those that were the most strongly correlated with the projected pre-XDR prevalence, indicating that pyrazinamide resistance acquired during first-line treatment subsequently promotes amplification to pre-XDR TB under pyrazinamide-containing second-line treatment. These findings suggest that the appropriate use of companion drugs may be critical to preventing the emergence of strains resistant to multiple core agents.
Keywords: antimicrobial combinations; mathematical modeling; multidrug resistance; pyrazinamide; tuberculosis.
Copyright © 2017 Fofana et al.
Figures



Comment in
-
Protecting Pyrazinamide, a Priority for Improving Outcomes in Multidrug-Resistant Tuberculosis Treatment.Antimicrob Agents Chemother. 2017 May 24;61(6):e00258-17. doi: 10.1128/AAC.00258-17. Print 2017 Jun. Antimicrob Agents Chemother. 2017. PMID: 28539498 Free PMC article. No abstract available.
-
Reply to Anthony et al., "Protecting Pyrazinamide, a Priority for Improving Outcomes in Multidrug-Resistant Tuberculosis Treatment".Antimicrob Agents Chemother. 2017 May 24;61(6):e00427-17. doi: 10.1128/AAC.00427-17. Print 2017 Jun. Antimicrob Agents Chemother. 2017. PMID: 28539499 Free PMC article. No abstract available.
References
-
- World Health Organization. 2014. Antimicrobial resistance: global report on surveillance 2014. World Health Organization, Geneva, Switzerland: http://www.who.int/drugresistance/documents/surveillancereport/en/ Accessed 29 June 2015.
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

