Transient receptor potential vanilloid 1-immunoreactive signals in murine enteric glial cells
- PMID: 27956799
- PMCID: PMC5124980
- DOI: 10.3748/wjg.v22.i44.9752
Transient receptor potential vanilloid 1-immunoreactive signals in murine enteric glial cells
Abstract
Aim: To investigate the possible involvement of transient receptor potential vanilloid 1 (TRPV1) in maturation of enteric glial cells (EGCs).
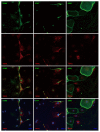
Methods: Immunohistochemical and immunocytochemical techniques were used to analyze EGC markers in myenteric plexus (MP) as well as cultured MP cells and EGCs using TRPV1 knockout (KO) mice.
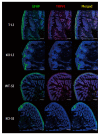
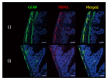
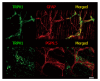
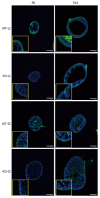
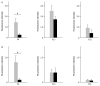
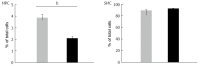
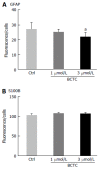
Results: We detected TRPV1-immunoreactive signals in EGC in the MP of wild-type (WT) but not KO mice. Expression of glial fibrillary acidic protein (GFAP) immunoreactive signals was lower at postnatal day (PD) 6 in KO mice, though the difference was not clear at PD 13 and PD 21. When MP cells were isolated and cultured from isolated longitudinal muscle-MP preparation from WT and KO mice, the yield of KO EGC was lower than that of WT EGC, while the yield of KO and WT smooth muscle cells showed no difference. Addition of BCTC, a TRPV1 antagonist, to enriched EGC culture resulted in a decrease in the protein ratio of GFAP to S100B, another EGC/astrocyte-specific marker.
Conclusion: These results address the possibility that TRPV1 may be involved in the maturation of EGC, though further studies are necessary to validate this possibility.
Keywords: Enteric glial cells; Enteric nervous system; Glial fibrillary acidic protein; S100B; Smooth muscle cells.
Conflict of interest statement
Conflict-of-interest statement: No potential conflicts of interest relevant to this article were reported.
Figures
Similar articles
-
Communication Between Enteric Neurons, Glia, and Nociceptors Underlies the Effects of Tachykinins on Neuroinflammation.Cell Mol Gastroenterol Hepatol. 2018 May 29;6(3):321-344. doi: 10.1016/j.jcmgh.2018.05.009. eCollection 2018. Cell Mol Gastroenterol Hepatol. 2018. PMID: 30116771 Free PMC article.
-
Expression profile of some neuronal and glial cell markers in the ovine ileal enteric nervous system during prenatal development.Acta Histochem. 2018 Nov;120(8):768-779. doi: 10.1016/j.acthis.2018.09.002. Epub 2018 Sep 11. Acta Histochem. 2018. PMID: 30217408
-
Characterization of human, mouse, and rat cultures of enteric glial cells and their effect on intestinal epithelial cells.Neurogastroenterol Motil. 2013 Nov;25(11):e755-64. doi: 10.1111/nmo.12200. Epub 2013 Aug 29. Neurogastroenterol Motil. 2013. PMID: 23991747
-
Enteric Glia: S100, GFAP, and Beyond.Anat Rec (Hoboken). 2019 Aug;302(8):1333-1344. doi: 10.1002/ar.24128. Epub 2019 Apr 29. Anat Rec (Hoboken). 2019. PMID: 30951262 Review.
-
S100B protein in the gut: the evidence for enteroglial-sustained intestinal inflammation.World J Gastroenterol. 2011 Mar 14;17(10):1261-6. doi: 10.3748/wjg.v17.i10.1261. World J Gastroenterol. 2011. PMID: 21455324 Free PMC article. Review.
Cited by
-
Cannabinoid and Cannabinoid-Related Receptors in the Myenteric Plexus of the Porcine Ileum.Animals (Basel). 2021 Jan 21;11(2):263. doi: 10.3390/ani11020263. Animals (Basel). 2021. PMID: 33494452 Free PMC article.
-
Role of Enteric Glia as Bridging Element between Gut Inflammation and Visceral Pain Consolidation during Acute Colitis in Rats.Biomedicines. 2021 Nov 12;9(11):1671. doi: 10.3390/biomedicines9111671. Biomedicines. 2021. PMID: 34829900 Free PMC article.
-
The Enteric Glia and Its Modulation by the Endocannabinoid System, a New Target for Cannabinoid-Based Nutraceuticals?Molecules. 2022 Oct 10;27(19):6773. doi: 10.3390/molecules27196773. Molecules. 2022. PMID: 36235308 Free PMC article. Review.
References
-
- Furness JB. The enteric nervous system and neurogastroenterology. Nat Rev Gastroenterol Hepatol. 2012;9:286–294. - PubMed
-
- Furness JB, Callaghan BP, Rivera LR, Cho HJ. The enteric nervous system and gastrointestinal innervation: integrated local and central control. Adv Exp Med Biol. 2014;817:39–71. - PubMed
-
- De Giorgio R, Giancola F, Boschetti E, Abdo H, Lardeux B, Neunlist M. Enteric glia and neuroprotection: basic and clinical aspects. Am J Physiol Gastrointest Liver Physiol. 2012;303:G887–G893. - PubMed
-
- Neunlist M, Rolli-Derkinderen M, Latorre R, Van Landeghem L, Coron E, Derkinderen P, De Giorgio R. Enteric glial cells: recent developments and future directions. Gastroenterology. 2014;147:1230–1237. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

