Development of new immunotherapy treatments in different cancer types
- PMID: 27974927
- PMCID: PMC5154307
Development of new immunotherapy treatments in different cancer types
Abstract
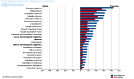
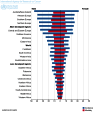
Cancer immunotherapy involves the use of therapeutic modalities that determine a manipulation of the immune system by using immune agents such as cytokines, vaccines, cell therapies and humoral, transfection agents. Immunotherapy of cancer has to stimulate the host's anti-tumor response by increasing the effector cell number and the production of soluble mediators and decrease the host's suppressor mechanisms by inducing tumor killing environment and by modulating immune checkpoints. Immunotherapy seems to work better in more immunogenic tumors. Making a review of literature, the article presents the new immunologic treatments in cancers less presented in the latest conferences, cancers in which, immunotherapy is still under investigation. Bladder cancer was the first indication for which immunotherapy was used in 1970. A promising clinical research in bladder cancer is the use of immune checkpoint inhibitors. Although breast cancer is considered immunologically silent, several preclinical and clinical studies suggested that immunotherapy has the potential to improve the clinical outcomes for patients with breast cancer. Cervical cancer, brain cancer, head and neck cancer and colorectal and esophageal cancers are cancer types for which new immune-based cancer treatments are currently under development. Recent agents used in clinical trials will be described in before mentioned cancers.
Keywords: arthrodesis; cancer; clinical studies; immunotherapy; new treatments.
Figures


References
-
- Zdimerova H, Albert ML, Ingersoll MA. Harnessing the Host Immune Response to Infection - BCG ImmunoTherapy for Bladder Cancer, Infection and Cancer: Bi-Directorial Interactions. :340–387.
-
- Rezaei N, Aalaei-Andabili SH, Kaufman HL. Introduction on Cancer Immunology and Immunotherapy. Cancer Immunology. 2014
-
- Sharma P. Bladder Cancer Immunotherapy review, Reviewed December 2014, Sources: ACS Facts and Figures 2014, National Cancer Institute Physician Data Query, National Cancer Institute Biological Therapies fact sheet, SEER Cancer Statistics Factsheets: Bladder Cancer, ClinicalTrials.gov, CRI documents
-
- "https://clinicaltrials.gov/show/NCT01928394 A Phase 1/ 2, Open-label Study of Nivolumab Monotherapy or Nivolumab Combined With Ipilimumab in Subjects With Advanced or Metastatic Solid Tumors, Bristol-Myers Squibb
-
- "https://clinicaltrials.gov/ct2/show/NCT02010203 A Phase 1/2 Study of HS-410 in Patients With Non-Muscle Invasive Bladder Cancer After TURBT, Heat Biologics
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
