Chiral-Substituted Poly-N-vinylpyrrolidinones and Bimetallic Nanoclusters in Catalytic Asymmetric Oxidation Reactions
- PMID: 27976875
- PMCID: PMC6394832
- DOI: 10.1021/jacs.6b12113
Chiral-Substituted Poly-N-vinylpyrrolidinones and Bimetallic Nanoclusters in Catalytic Asymmetric Oxidation Reactions
Abstract
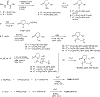
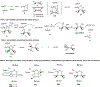
A new class of poly-N-vinylpyrrolidinones containing an asymmetric center at C5 of the pyrrolidinone ring were synthesized from l-amino acids. The polymers, particularly 17, were used to stabilize nanoclusters such as Pd/Au for the catalytic asymmetric oxidations of 1,3- and 1,2-cycloalkanediols and alkenes, and Cu/Au was used for C-H oxidation of cycloalkanes. It was found that the bulkier the C5 substituent in the pyrrolidinone ring, the greater the optical yields produced. Both oxidative kinetic resolution of (±)-1,3- and 1,2-trans-cycloalkanediols and desymmetrization of meso cis-diols took place with 0.15 mol % Pd/Au (3:1)-17 under oxygen atmosphere in water to give excellent chemical and optical yields of (S)-hydroxy ketones. Various alkenes were oxidized with 0.5 mol % Pd/Au (3:1)-17 under 30 psi of oxygen in water to give the dihydroxylated products in >93% ee. Oxidation of (R)-limonene at 25 °C occurred at the C-1,2-cyclic alkene function yielding (1S,2R,4R)-dihydroxylimonene 49 in 92% yield. Importantly, cycloalkanes were oxidized with 1 mol % Cu/Au (3:1)-17 and 30% H2O2 in acetonitrile to afford chiral ketones in very good to excellent chemical and optical yields. Alkene function was not oxidized under the reaction conditions. Mechanisms were proposed for the oxidation reactions, and observed stereo- and regio-chemistry were summarized.
Conflict of interest statement
Notes
The authors declare no competing financial interest.
Figures


Similar articles
-
Synthesis and Characterization of Bimetallic Nanoclusters Stabilized by Chiral and Achiral Polyvinylpyrrolidinones. Catalytic C(sp3)-H Oxidation.J Org Chem. 2022 May 20;87(10):6742-6759. doi: 10.1021/acs.joc.2c00449. Epub 2022 May 5. J Org Chem. 2022. PMID: 35511477 Free PMC article.
-
Synthetic applications of nonmetal catalysts for homogeneous oxidations.Chem Rev. 2001 Nov;101(11):3499-548. doi: 10.1021/cr000019k. Chem Rev. 2001. PMID: 11840992
-
Indication of Pd-C or Cu-C Intermediate in Respective Bimetallic Nanoclusters Pd/Au-PVP or Cu/Au-PVP Catalyzed Oxidations of endo-4-Oxatricyclo[5.2.1.02,6]-8-decene and Tetrahydro-γ-carbolines.Synthesis (Stuttg). 2023 Apr;55(8):1227-1240. doi: 10.1055/a-2001-6888. Epub 2020 Dec 20. Synthesis (Stuttg). 2023. PMID: 37151600 Free PMC article.
-
Metal-catalyzed asymmetric oxidations.Med Res Rev. 1999 Sep;19(5):348-56. doi: 10.1002/(sici)1098-1128(199909)19:5<348::aid-med3>3.0.co;2-u. Med Res Rev. 1999. PMID: 10502739 Review.
-
Catalytic enantioselective synthesis of chiral organic compounds of ultra-high purity of >99% ee.Proc Jpn Acad Ser B Phys Biol Sci. 2015;91(8):369-93. doi: 10.2183/pjab.91.369. Proc Jpn Acad Ser B Phys Biol Sci. 2015. PMID: 26460317 Free PMC article. Review.
Cited by
-
Diastereoselective desymmetric 1,2-cis-glycosylation of meso-diols via chirality transfer from a glycosyl donor.Nat Commun. 2020 May 15;11(1):2431. doi: 10.1038/s41467-020-16365-8. Nat Commun. 2020. PMID: 32415161 Free PMC article.
-
Synthesis and migrastatic activity of cytochalasin analogues lacking a macrocyclic moiety.RSC Med Chem. 2023 Nov 28;15(1):322-343. doi: 10.1039/d3md00535f. eCollection 2024 Jan 25. RSC Med Chem. 2023. PMID: 38283219 Free PMC article.
-
Synthesis of Composition-Tunable Ag-Cu Bimetallic Nanoparticles Through Plasma-Driven Solution Electrolysis.Nanomaterials (Basel). 2024 Oct 31;14(21):1758. doi: 10.3390/nano14211758. Nanomaterials (Basel). 2024. PMID: 39513838 Free PMC article.
-
Strategies for Accessing cis-1-Amino-2-Indanol.Molecules. 2024 May 22;29(11):2442. doi: 10.3390/molecules29112442. Molecules. 2024. PMID: 38893318 Free PMC article. Review.
-
Systematic synthetic study of four diastereomerically distinct limonene-1,2-diols and their corresponding cyclic carbonates.Beilstein J Org Chem. 2019 Jan 14;15:130-136. doi: 10.3762/bjoc.15.13. eCollection 2019. Beilstein J Org Chem. 2019. PMID: 30745988 Free PMC article.
References
-
- Zhang H; Watanabe T; Okumura M; Haruta M; Toshima N Nature Mater 2012, 11, 49. - PubMed
-
- Miyamura H; Kobayashi S Acc. Chem. Res 2014, 47, 1054. - PubMed
-
- Hou W; Dehm NA; Scott RWJ J. Catal 2008, 253, 22.
-
- Toshima N; Yonezawa T New J. Chem 1998, 22, 1179.
-
- Yao H; Kobayashi R Colloid Interface Sci 2014, 419, 1. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

