The Development of Spasmolytic Polypeptide/TFF2-Expressing Metaplasia (SPEM) During Gastric Repair Is Absent in the Aged Stomach
- PMID: 27990460
- PMCID: PMC5042762
- DOI: 10.1016/j.jcmgh.2016.05.004
The Development of Spasmolytic Polypeptide/TFF2-Expressing Metaplasia (SPEM) During Gastric Repair Is Absent in the Aged Stomach
Abstract
Background & aims: During aging, physiological changes in the stomach result in more tenuous gastric tissue that is less capable of repairing injury, leading to increased susceptibility to chronic ulceration. Spasmolytic polypeptide/trefoil factor 2-expressing metaplasia (SPEM) is known to emerge after parietal cell loss and during Helicobacter pylori infection, however, its role in gastric ulcer repair is unknown. Therefore, we sought to investigate if SPEM plays a role in epithelial regeneration.
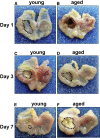
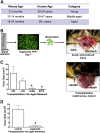
Methods: Acetic acid ulcers were induced in young (2-3 mo) and aged (18-24 mo) C57BL/6 mice to determine the quality of ulcer repair with advancing age. Yellow chameleon 3.0 mice were used to generate yellow fluorescent protein-expressing organoids for transplantation. Yellow fluorescent protein-positive gastric organoids were transplanted into the submucosa and lumen of the stomach immediately after ulcer induction. Gastric tissue was collected and analyzed to determine the engraftment of organoid-derived cells within the regenerating epithelium.
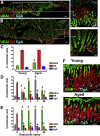
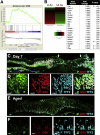
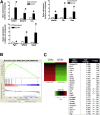
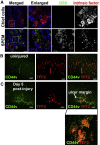
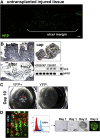
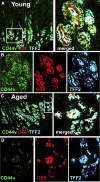
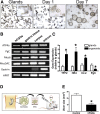
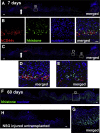
Results: Wound healing in young mice coincided with the emergence of SPEM within the ulcerated region, a response that was absent in the aged stomach. Although aged mice showed less metaplasia surrounding the ulcerated tissue, organoid-transplanted aged mice showed regenerated gastric glands containing organoid-derived cells. Organoid transplantation in the aged mice led to the emergence of SPEM and gastric regeneration.
Conclusions: These data show the development of SPEM during gastric repair in response to injury that is absent in the aged stomach. In addition, gastric organoids in an injury/transplantation mouse model promoted gastric regeneration.
Keywords: CD44v; CD44v, variant isoform of CD44; Cftr, cystic fibrosis transmembrane conductance regulator; CgA, chromagranin A; Clu, Clusterin; Ctss, cathepsin S; DMEM, Dulbecco's modified Eagle medium; DPBS, Dulbecco's phosphate buffered saline; Dmbt1, deleted in malignant brain tumors 1; ES, enrichment score; Epithelial Regeneration; GSEA, gene set enrichment analysis; GSII, Griffonia simplicifolia II; Gastric Cancer; Gpx2, glutathione peroxidase 2 (gastrointestinal); HK, hydrogen potassium adenosine triphosphatase; Human Gastric Organoids; IF, intrinsic factor; Mad2I1, MAD2 mitotic arrest deficient-like 1; Mmp12, matrix metallopeptidase 12 (macrophage elastase); PBS, phosphate-buffered saline; SPEM, spasmolytic polypeptide expressing metaplasia; TFF, trefoil factor; TX, Triton X-100 in PBS; UEA1, ulex europaeus; Wfdc2, WAP 4-disulfide core domain 2; YFP, yellow fluorescent protein; hFGO, human-derived fundic gastric organoid; qRT-PCR, quantitative reverse-transcription polymerase chain reaction.
Figures




References
-
- Salles N. Is stomach spontaneously ageing? Pathophysiology of the ageing stomach. Best Pract Res Clin Gastroenterol. 2009;23:805–819. - PubMed
-
- Ahluwalia A., Jones M.K., Deng X. An imbalance between VEGF and endostatin underlies impaired angiogenesis in gastric mucosa of aging rats. Am J Physiol Gastrointest Liver Physiol. 2013;305:G325–G332. - PubMed
-
- Ahluwalia A., Jones M.K., Szabo S. Aging impairs transcriptional regulation of vascular endothelial growth factor in human microvascular endothelial cells: implications for angiogenesis and cell survival. J Physiol Pharmacol. 2014;65:209–215. - PubMed
-
- Tarnawski A.S., Ahluwalia A., Jones M.K. Angiogenesis in gastric mucosa: an important component of gastric erosion and ulcer healing and its impairment in aging. J Gastroenterol Hepatol. 2014;29:112–123. - PubMed
-
- Jones J.I., Hawkey C.J. Physiology and organ-related pathology of the elderly: stomach ulcers. Best Pract Res Clin Gastroenterol. 2001;15:943–961. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

