Identification and Characterization of Roseltide, a Knottin-type Neutrophil Elastase Inhibitor Derived from Hibiscus sabdariffa
- PMID: 27991569
- PMCID: PMC5171801
- DOI: 10.1038/srep39401
Identification and Characterization of Roseltide, a Knottin-type Neutrophil Elastase Inhibitor Derived from Hibiscus sabdariffa
Abstract
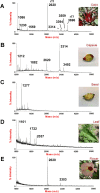
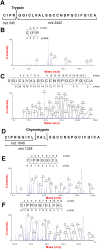
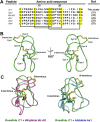
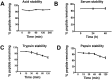
Plant knottins are of therapeutic interest due to their high metabolic stability and inhibitory activity against proteinases involved in human diseases. The only knottin-type proteinase inhibitor against porcine pancreatic elastase was first identified from the squash family in 1989. Here, we report the identification and characterization of a knottin-type human neutrophil elastase inhibitor from Hibiscus sabdariffa of the Malvaceae family. Combining proteomic and transcriptomic methods, we identified a panel of novel cysteine-rich peptides, roseltides (rT1-rT8), which range from 27 to 39 residues with six conserved cysteine residues. The 27-residue roseltide rT1 contains a cysteine spacing and amino acid sequence that is different from the squash knottin-type elastase inhibitor. NMR analysis demonstrated that roseltide rT1 adopts a cystine-knot fold. Transcriptome analyses suggested that roseltides are bioprocessed by asparagine endopeptidases from a three-domain precursor. The cystine-knot structure of roseltide rT1 confers its high resistance against degradation by endopeptidases, 0.2 N HCl, and human serum. Roseltide rT1 was shown to inhibit human neutrophil elastase using enzymatic and pull-down assays. Additionally, roseltide rT1 ameliorates neutrophil elastase-stimulated cAMP accumulation in vitro. Taken together, our findings demonstrate that roseltide rT1 is a novel knottin-type neutrophil elastase inhibitor with therapeutic potential for neutrophil elastase associated diseases.
Figures

Similar articles
-
Plant-derived mitochondria-targeting cysteine-rich peptide modulates cellular bioenergetics.J Biol Chem. 2019 Mar 15;294(11):4000-4011. doi: 10.1074/jbc.RA118.006693. Epub 2019 Jan 23. J Biol Chem. 2019. PMID: 30674551 Free PMC article.
-
Roseltide rT7 is a disulfide-rich, anionic, and cell-penetrating peptide that inhibits proteasomal degradation.J Biol Chem. 2019 Dec 20;294(51):19604-19615. doi: 10.1074/jbc.RA119.010796. Epub 2019 Nov 14. J Biol Chem. 2019. PMID: 31727740 Free PMC article.
-
Hydoroxyhibiscone A, a novel human neutrophil elastase inhibitor from Hibiscus syriacus.J Microbiol Biotechnol. 2010 Aug;20(8):1189-91. doi: 10.4014/jmb.1004.04028. J Microbiol Biotechnol. 2010. PMID: 20798580
-
Natural compounds as inhibitors of human neutrophil elastase.Planta Med. 2007 May;73(5):401-20. doi: 10.1055/s-2007-967183. Epub 2007 Apr 20. Planta Med. 2007. PMID: 17447201 Review.
-
Natural Phenolics as Inhibitors of the Human Neutrophil Elastase (HNE) Release: An Overview of Natural Anti-inflammatory Discoveries during Recent Years.Antiinflamm Antiallergy Agents Med Chem. 2018;17(2):70-94. doi: 10.2174/1871523017666180910104946. Antiinflamm Antiallergy Agents Med Chem. 2018. PMID: 30198444 Review.
Cited by
-
Recent Advances in Chemical Biology of Mitochondria Targeting.Front Chem. 2021 May 3;9:683220. doi: 10.3389/fchem.2021.683220. eCollection 2021. Front Chem. 2021. PMID: 34012953 Free PMC article. Review.
-
Peptide Human Neutrophil Elastase Inhibitors from Natural Sources: An Overview.Int J Mol Sci. 2022 Mar 8;23(6):2924. doi: 10.3390/ijms23062924. Int J Mol Sci. 2022. PMID: 35328340 Free PMC article. Review.
-
Ginsentides: Cysteine and Glycine-rich Peptides from the Ginseng Family with Unusual Disulfide Connectivity.Sci Rep. 2018 Nov 1;8(1):16201. doi: 10.1038/s41598-018-33894-x. Sci Rep. 2018. PMID: 30385768 Free PMC article.
-
Hololectin Interdomain Linker Determines Asparaginyl Endopeptidase-Mediated Maturation of Antifungal Hevein-Like Peptides in Oats.Front Plant Sci. 2022 May 10;13:899740. doi: 10.3389/fpls.2022.899740. eCollection 2022. Front Plant Sci. 2022. PMID: 35620686 Free PMC article.
-
In Vitro Antiprotozoal Activity of Hibiscus sabdariffa Extract against a Ciliate Causing High Mortalities in Turbot Aquaculture.Biology (Basel). 2023 Jun 26;12(7):912. doi: 10.3390/biology12070912. Biology (Basel). 2023. PMID: 37508344 Free PMC article.
References
-
- Ali B. H., Al Wabel N. & Blunden G. Phytochemical, pharmacological and toxicological aspects of Hibiscus sabdariffa L.: a review. Phytother. Res. 19, 369–375 (2005). - PubMed
-
- Wilson F. D. & Menzel M. Y. Kenaf (Hibiscus cannabinus), roselle (Hibiscus sabdariffa). Econ. Bot. 18, 80–91 (1964).
-
- Herrera-Arellano A. et al.. Clinical effects produced by a standardized herbal medicinal product of Hibiscus sabdariffa on patients with hypertension. A randomized, double-blind, lisinopril-controlled clinical trial. Planta Med. 73, 6–12 (2007). - PubMed
-
- Ajay M., Chai H. J., Mustafa A. M., Gilani A. H. & Mustafa M. R. Mechanisms of the anti-hypertensive effect of Hibiscus sabdariffa L. calyces. J. Ethnopharmacol. 109, 388–393 (2007). - PubMed
-
- Chen C. C. et al.. Hibiscus sabdariffa extract inhibits the development of atherosclerosis in cholesterol-fed rabbits. J. Agric. Food Chem. 51, 5472–5477 (2003). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

