Octreotide LAR and Prednisone as Neoadjuvant Treatment in Patients with Primary or Locally Recurrent Unresectable Thymic Tumors: A Phase II Study
- PMID: 27992479
- PMCID: PMC5161359
- DOI: 10.1371/journal.pone.0168215
Octreotide LAR and Prednisone as Neoadjuvant Treatment in Patients with Primary or Locally Recurrent Unresectable Thymic Tumors: A Phase II Study
Abstract
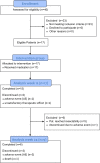
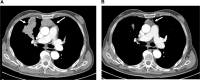
Therapeutic options to cure advanced, recurrent, and unresectable thymomas are limited. The most important factor for long-term survival of thymoma patients is complete resection (R0) of the tumor. We therefore evaluated the response to and the induction of resectability of primarily or locally recurrent unresectable thymomas and thymic carcinomas by octreotide Long-Acting Release (LAR) plus prednisone therapy in patients with positive octreotide scans. In this open label, single-arm phase II study, 17 patients with thymomas considered unresectable or locally recurrent thymoma (n = 15) and thymic carcinoma (n = 2) at Masaoka stage III were enrolled. Octreotide LAR (30 mg once every 2 weeks) was administered in combination with prednisone (0.6 mg/kg per day) for a maximum of 24 weeks (study design according to Fleming´s one sample multiple testing procedure for phase II clinical trials). Tumor size was evaluated by volumetric CT measurements, and a decrease in tumor volume of at least 20% at week 12 compared to baseline was considered as a response. We found that octreotide LAR plus prednisone elicited response in 15 of 17 patients (88%). Median reduction of tumor volume after 12 weeks of treatment was 51% (range 20%-86%). Subsequently, complete surgical resection was achieved in five (29%) and four patients (23%) after 12 and 24 weeks, respectively. Octreotide LAR plus prednisone treatment was discontinued in two patients before week 12 due to unsatisfactory therapeutic effects or adverse events. The most frequent adverse events were gastrointestinal (71%), infectious (65%), and hematological (41%) complications. In conclusion, octreotide LAR plus prednisone is efficacious in patients with primary or recurrent unresectable thymoma with respect to tumor regression. Octreotide LAR plus prednisone was well tolerated and adverse events were in line with the known safety profile of both agents.
Conflict of interest statement
Dr. Kleylein-Sohn, Dr. May and Dr. Baierlein are employees of Novartis. Dr. Boy is an employee of Asclepios clinic. Prof. Schalke reports grants from Novartis within and outside the submitted work. This does not alter our adherence to PLOS ONE policies on sharing data and materials. The other authors declare no conflicts of interest.
Figures

References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

