Efficacy and safety of adult human bone marrow-derived, cultured, pooled, allogeneic mesenchymal stromal cells (Stempeucel®): preclinical and clinical trial in osteoarthritis of the knee joint
- PMID: 27993154
- PMCID: PMC5168586
- DOI: 10.1186/s13075-016-1195-7
Efficacy and safety of adult human bone marrow-derived, cultured, pooled, allogeneic mesenchymal stromal cells (Stempeucel®): preclinical and clinical trial in osteoarthritis of the knee joint
Abstract
Background: Osteoarthritis (OA) is a common and debilitating chronic degenerative disease of the joints. Currently, cell-based therapy is being explored to address the repair of damaged articular cartilage in the knee joint.
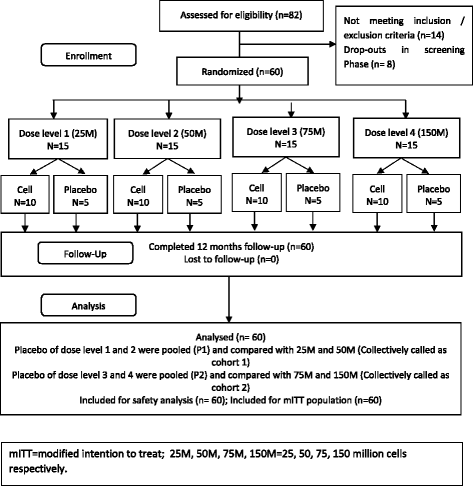
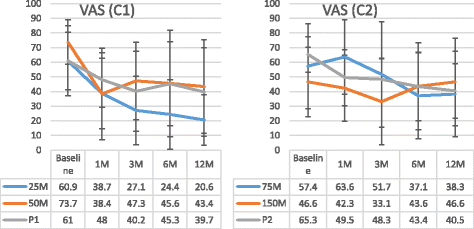
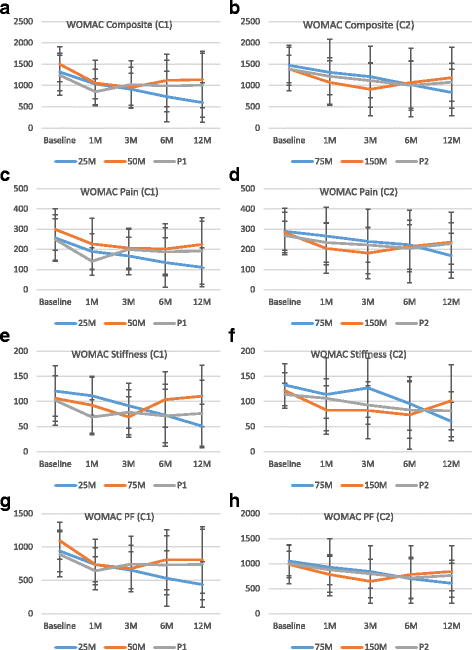
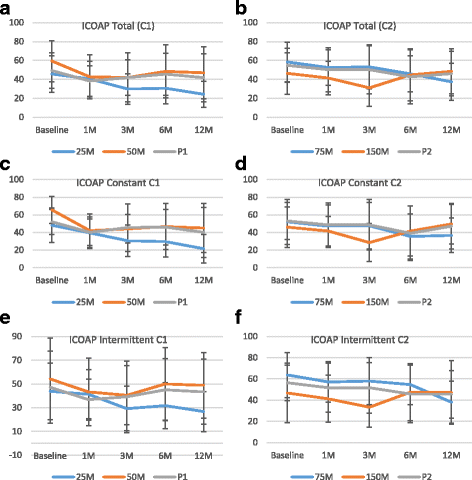
Methods: The in vitro differentiation potential of adult human bone marrow-derived, cultured, pooled, allogeneic mesenchymal stromal cells (Stempeucel®) was determined by differentiating the cells toward the chondrogenic lineage and quantifying sulfated glycosaminoglycan (sGAG). The mono-iodoacetate (MIA)-induced preclinical model of OA has been used to demonstrate pain reduction and cartilage formation. In the clinical study, 60 OA patients were randomized to receive different doses of cells (25, 50, 75, or 150 million cells) or placebo. Stempeucel® was administered by intra-articular (IA) injection into the knee joint, followed by 2 ml hyaluronic acid (20 mg). Subjective evaluations-visual analog scale (VAS) for pain, intermittent and constant osteoarthritis pain (ICOAP), and Western Ontario and McMaster Universities Osteoarthritis (WOMAC-OA) index-were performed at baseline and at 1, 3, 6, and 12 months of follow-up. Magnetic resonance imaging of the knee was performed at baseline, and at 6 and 12 months follow-up for cartilage evaluation.
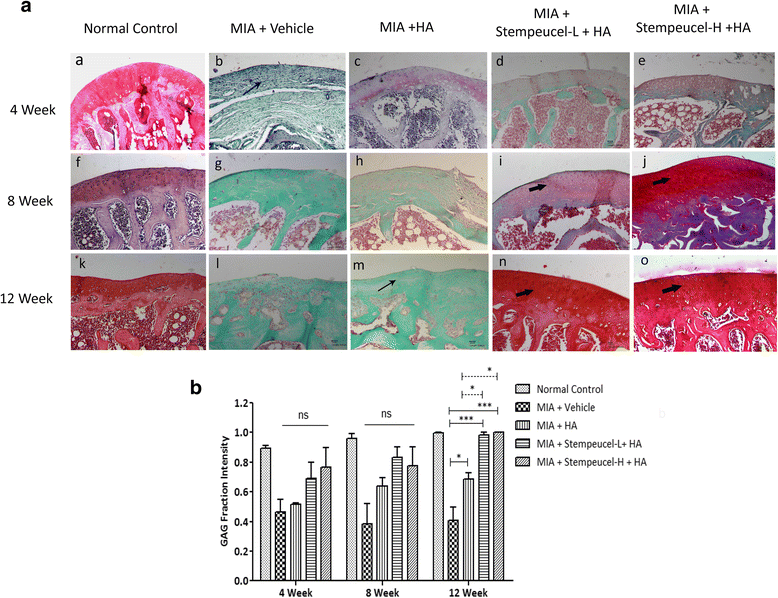
Results: Stempeucel® differentiated into the chondrogenic lineage in vitro with downregulation of Sox9 and upregulation of Col2A genes. Furthermore, Stempeucel® differentiated into chondrocytes and synthesized a significant amount of sGAG (30 ± 1.8 μg/μg GAG/DNA). In the preclinical model of OA, Stempeucel® reduced pain significantly and also repaired damaged articular cartilage in rats. In the clinical study, IA administration of Stempeucel® was safe, and a trend towards improvement was seen in the 25-million-cell dose group in all subjective parameters (VAS, ICOAP, andWOMAC-OA scores), although this was not statistically significant when compared to placebo. Adverse events were predominant in the higher dose groups (50, 75, and 150 million cells). Knee pain and swelling were the most common adverse events. The whole-organ magnetic resonance imaging score of the knee did not reveal any difference from baseline and the placebo group.
Conclusion: Intra-articular administration of Stempeucel® is safe. A twenty-five-million-cell dose may be the most effective among the doses tested for pain reduction. Clinical studies with a larger patient population are required to demonstrate a robust therapeutic efficacy of Stempeucel® in OA.
Trial registration: Clinicaltrials.gov NCT01453738 . Registered 13 October 2011.
Keywords: Cell therapy; Mesenchymal stromal cells; Osteoarthritis.
Figures


References
-
- Murray C, Lopez A. The global burden of disease: a comprehensive assessment of mortality and disability from disease, injuries and risk factors in 1990 and projected to 2020. Boston: Harvard University Press; 1996.
-
- Osteoarthritis: care and management. https://www.nice.org.uk/guidance/cg177. Accessed 8 May 2016.
-
- Hochberg MC, Altman RD, April KT, Benkhalti M, Guyatt G, McGowan J, et al. American College of Rheumatology 2012 recommendations for the use of nonpharmacologic and pharmacologic therapies in osteoarthritis of the hand, hip, and knee. Arthritis Care Res. 2012;64(4):465–474. doi: 10.1002/acr.21596. - DOI - PubMed
-
- Jordan KM, Arden NK, Doherty M, Bannwarth B, Bijlsma JWJ, Dieppe P, et al. EULAR Recommendations 2003: an evidence based approach to the management of knee osteoarthritis: report of a Task Force of the Standing Committee for International Clinical Studies Including Therapeutic Trials (ESCISIT) Ann Rheum Dis. 2003;62(12):1145–1155. doi: 10.1136/ard.2003.011742. - DOI - PMC - PubMed
-
- Schrama JC, Espehaug B, Hallan G, Engesæter LB, Furnes O, Havelin LI, et al. Risk of revision for infection in primary total hip and knee arthroplasty in patients with rheumatoid arthritis compared with osteoarthritis: a prospective, population-based study on 108,786 hip and knee joint arthroplasties from the Norwegian Arthroplasty Register. Arthritis Care Res. 2010;62(4):473–479. doi: 10.1002/acr.20036. - DOI - PubMed
Publication types
MeSH terms
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

