Roles of GSK-3 and microRNAs on epithelial mesenchymal transition and cancer stem cells
- PMID: 27999207
- PMCID: PMC5355173
- DOI: 10.18632/oncotarget.13991
Roles of GSK-3 and microRNAs on epithelial mesenchymal transition and cancer stem cells
Abstract
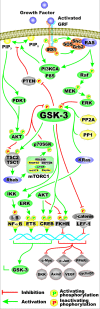
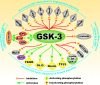
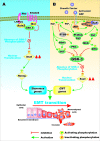
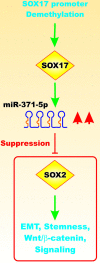
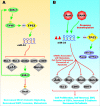
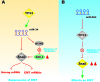
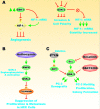
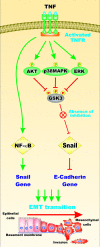
Various signaling pathways exert critical roles in the epithelial to mesenchymal transition (EMT) and cancer stem cells (CSCs). The Wnt/beta-catenin, PI3K/PTEN/Akt/mTORC, Ras/Raf/MEK/ERK, hedgehog (Hh), Notch and TP53 pathways elicit essential regulatory influences on cancer initiation, EMT and progression. A common kinase involved in all these pathways is moon-lighting kinase glycogen synthase kinase-3 (GSK-3). These pathways are also regulated by micro-RNAs (miRs). TP53 and components of these pathways can regulate the expression of miRs. Targeting members of these pathways may improve cancer therapy in those malignancies that display their abnormal regulation. This review will discuss the interactions of the multi-functional GSK-3 enzyme in the Wnt/beta-catenin, PI3K/PTEN/Akt/mTORC, Ras/Raf/MEK/ERK, Hh, Notch and TP53 pathways. The regulation of these pathways by miRs and their effects on CSC generation, EMT, invasion and metastasis will be discussed.
Keywords: Akt; GSK-3; PI3K; Wnt/beta-catenin; cancer stem cells.
Conflict of interest statement
The authors declare that they have no conflicts of interest with publication of this manuscript.
Figures




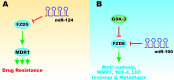
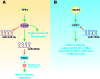

References
-
- McCubrey JA, Davis NM, Abrams SL, Montalto G, Cervello M, Basecke J, Libra M, Nicoletti F, Cocco L, Martelli AM, Steelman LS. Diverse roles of GSK-3: Tumor promoter-tumor suppressor, target in cancer therapy. Advances in biological regulation. 2014;54:176–196. - PubMed
-
- McCubrey JA, Steelman LS, Bertrand FE, Davis NM, Abrams SL, Montalto G, D'Assoro AB, Libra M, Nicoletti F, Maestro R, Basecke J, Cocco L, Cervello M, et al. Multifaceted roles of GSK-3 and Wnt/β-catenin in hematopoiesis and leukemogenesis: opportunities for therapeutic intervention. Leukemia. 2014;28:15–33. - PMC - PubMed
-
- McCubrey JA, Steelman LS, Bertrand FE, Davis NM, Sokolosky M, Abrams SL, Montalto G, D'Assoro AB, Libra M, Nicoletti F, Maestro R, Basecke J, Rakus D, et al. GSK-3 as potential target for therapeutic intervention in cancer. Oncotarget. 2014;5:2881–2911. doi: 10.18632/oncotarget.2037. - DOI - PMC - PubMed
-
- McCubrey JA, Rakus D, Gizak A, Steelman LS, Abrams SL, Lertpiriyapong K, Fitzgerald TL, Yang LV, Montalto G, Cervello M, Libra M, Nicoletti F, Scalisi A, et al. Effects of mutations in Wnt/β-catenin, hedgehog, Notch and PI3K pathways on GSK-3 activity-Diverse effects on cell growth, metabolism and cancer. Biochim Biophys Acta. 2016;1863:2942–2976. - PubMed
-
- Davis NM, Sokolosky M, Stadelman K, Abrams SL, Libra M, Candido S, Nicoletti F, Polesel J, Maestro R, D'Assoro A, Drobot L, Rakus D, Gizak A, et al. Deregulation of the EGFR/PI3K/PTEN/Akt/mTORC1 pathway in breast cancer: possibilities for therapeutic intervention. Oncotarget. 2014;5:4603–4650. doi: 10.18632/oncotarget.2209. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

