Generation of Human Corneal Endothelial Cells via In Vitro Ocular Lineage Restriction of Pluripotent Stem Cells
- PMID: 28002562
- PMCID: PMC5215466
- DOI: 10.1167/iovs.16-20024
Generation of Human Corneal Endothelial Cells via In Vitro Ocular Lineage Restriction of Pluripotent Stem Cells
Abstract
Purpose: We generate a renewable supply of corneal endothelial cells (CEC) from human pluripotent stem cells (PSCs) under defined culture conditions.
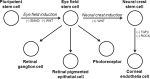
Methods: Corneal endothelial cell induction was driven by small molecules in a stepwise fashion of lineage specification. During the initial phase, PSC fate was restricted to the eye field-like state and became eye field stem cells (EFSCs). In the second phase, PSC-derived EFSCs were further directed toward either neural crest lineage or retinal lineage. The CECs were directly induced from ocular neural crest stem cells (NCSCs) by suppressing TGF-β and ROCK signaling.
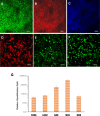
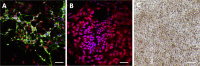
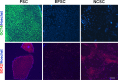
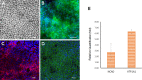
Results: Under chemically defined conditions, PSCs were massively converted into EFSCs and subsequently NCSCs. Eye field cell identity was characterized by the expression of key fate restriction factors for early eye field cells, such as PAX6, LHX2, and VSX2. The induction of ocular NCSCs was initiated by promoting WNT signaling in EFSCs. Within 2 weeks of induction, the majority of cells expressed the typical neural crest markers p75NTR and HNK-1. Eye field stem cell-derived NCSCs can be propagated and cryopreserved. Subsequently, a CEC monolayer was induced from adherent NCSCs in the presence of small molecular inhibitors to suppress TGF-β and ROCK signaling. The polygon-shaped CEC-like cells became visible after a week in culture. The NCSC-derived CECs expressed typical CEC markers, such as N-Cadherin and Na+/K+-ATPase.
Conclusions: A novel small molecule-based approach was developed to derive human CECs from PSCs via ocular lineage specification. Moreover, EFSC-derived NCSCs could serve as an immediate source cell for rapid CEC induction in vitro.
Figures

Similar articles
-
Guttae Morphology After Cultured Corneal Endothelial Cell Transplant in Fuchs Endothelial Corneal Dystrophy.JAMA Ophthalmol. 2024 Sep 1;142(9):818-826. doi: 10.1001/jamaophthalmol.2024.2718. JAMA Ophthalmol. 2024. PMID: 39052247 Free PMC article.
-
TGF-β Promotes Endothelial-to-Mesenchymal Transition and Alters Corneal Endothelial Cell Migration in Fuchs Endothelial Corneal Dystrophy.Int J Mol Sci. 2025 Jul 11;26(14):6685. doi: 10.3390/ijms26146685. Int J Mol Sci. 2025. PMID: 40724935 Free PMC article.
-
The PERK-p38 MAPK Axis Drives Endoplasmic Reticulum Stress-Induced Apoptosis in Fuchs Endothelial Corneal Dystrophy.Invest Ophthalmol Vis Sci. 2025 Aug 1;66(11):63. doi: 10.1167/iovs.66.11.63. Invest Ophthalmol Vis Sci. 2025. PMID: 40856649
-
Preparation of astrocytes by directed differentiation of pluripotent stem cells and somatic cell transdifferentiation.Dev Neurobiol. 2023 Oct-Nov;83(7-8):282-292. doi: 10.1002/dneu.22929. Epub 2023 Oct 3. Dev Neurobiol. 2023. PMID: 37789524 Review.
-
Hematopoietic stem cells and HSCs-derived from pluripotent stem cells: a hopeful outlook in cell-based therapeutics.Mol Biol Rep. 2025 Jun 30;52(1):657. doi: 10.1007/s11033-025-10730-w. Mol Biol Rep. 2025. PMID: 40587040 Review.
Cited by
-
A Guide to the Development of Human CorneaOrganoids from Induced Pluripotent Stem Cells in Culture.Methods Mol Biol. 2020;2145:51-58. doi: 10.1007/978-1-0716-0599-8_5. Methods Mol Biol. 2020. PMID: 32542600 Free PMC article.
-
Induction of Corneal Endothelial-like Cells from Mesenchymal Stem Cells of the Umbilical Cord.Int J Mol Sci. 2022 Dec 6;23(23):15408. doi: 10.3390/ijms232315408. Int J Mol Sci. 2022. PMID: 36499735 Free PMC article.
-
Differential Susceptibility of Fetal Retinal Pigment Epithelial Cells, hiPSC- Retinal Stem Cells, and Retinal Organoids to Zika Virus Infection.Viruses. 2023 Jan 1;15(1):142. doi: 10.3390/v15010142. Viruses. 2023. PMID: 36680182 Free PMC article.
-
Corneal Regeneration Using Adipose-Derived Mesenchymal Stem Cells.Cells. 2022 Aug 16;11(16):2549. doi: 10.3390/cells11162549. Cells. 2022. PMID: 36010626 Free PMC article. Review.
-
Long-term corneal recovery by simultaneous delivery of hPSC-derived corneal endothelial precursors and nicotinamide.J Clin Invest. 2022 Jan 4;132(1):e146658. doi: 10.1172/JCI146658. J Clin Invest. 2022. PMID: 34981789 Free PMC article.
References
-
- Bourne WM,, Nelson LR,, Hodge DO. Central corneal endothelial cell changes over a ten-year period. Invest Ophthalmol Vis Sci. 1997; 38: 779–782. - PubMed
-
- Joyce NC,, Harris DL,, Mello DM. Mechanisms of mitotic inhibition in corneal endothelium: contact inhibition and TGF-beta2. Invest Ophthalmol Vis Sci. 2002; 43: 2152–2159. - PubMed
-
- Ishino Y,, Sano Y,, Nakamura T,, et al. Amniotic membrane as a carrier for cultivated human corneal endothelial cell transplantation. Invest Ophthalmol Vis Sci. 2004; 45: 800–806. - PubMed
-
- Joyce NC. Proliferative capacity of the corneal endothelium. Prog Retin Eye Res. 2003; 22: 359–389. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

