Whole-GUV patch-clamping
- PMID: 28003462
- PMCID: PMC5240670
- DOI: 10.1073/pnas.1609142114
Whole-GUV patch-clamping
Abstract
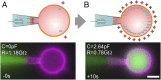
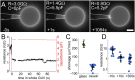
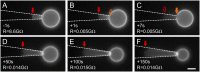
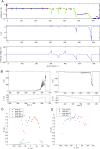
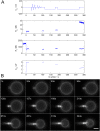
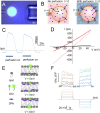
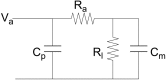
Studying how the membrane modulates ion channel and transporter activity is challenging because cells actively regulate membrane properties, whereas existing in vitro systems have limitations, such as residual solvent and unphysiologically high membrane tension. Cell-sized giant unilamellar vesicles (GUVs) would be ideal for in vitro electrophysiology, but efforts to measure the membrane current of intact GUVs have been unsuccessful. In this work, two challenges for obtaining the "whole-GUV" patch-clamp configuration were identified and resolved. First, unless the patch pipette and GUV pressures are precisely matched in the GUV-attached configuration, breaking the patch membrane also ruptures the GUV. Second, GUVs shrink irreversibly because the membrane/glass adhesion creating the high-resistance seal (>1 GΩ) continuously pulls membrane into the pipette. In contrast, for cell-derived giant plasma membrane vesicles (GPMVs), breaking the patch membrane allows the GPMV contents to passivate the pipette surface, thereby dynamically blocking membrane spreading in the whole-GMPV mode. To mimic this dynamic passivation mechanism, beta-casein was encapsulated into GUVs, yielding a stable, high-resistance, whole-GUV configuration for a range of membrane compositions. Specific membrane capacitance measurements confirmed that the membranes were truly solvent-free and that membrane tension could be controlled over a physiological range. Finally, the potential for ion transport studies was tested using the model ion channel, gramicidin, and voltage-clamp fluorometry measurements were performed with a voltage-dependent fluorophore/quencher pair. Whole-GUV patch-clamping allows ion transport and other voltage-dependent processes to be studied while controlling membrane composition, tension, and shape.
Keywords: biomimetic system; electrophysiology; giant unilamellar vesicle; lipid–glass interaction; patch clamp.
Conflict of interest statement
The authors declare no conflict of interest.
Figures






Similar articles
-
Single GUV method reveals interaction of tea catechin (-)-epigallocatechin gallate with lipid membranes.Biophys J. 2007 May 1;92(9):3178-94. doi: 10.1529/biophysj.106.097105. Epub 2007 Feb 9. Biophys J. 2007. PMID: 17293394 Free PMC article.
-
Ultrathin shell double emulsion templated giant unilamellar lipid vesicles with controlled microdomain formation.Small. 2014 Mar 12;10(5):950-6. doi: 10.1002/smll.201301904. Epub 2013 Oct 22. Small. 2014. PMID: 24150883
-
Functional reconstitution of a voltage-gated potassium channel in giant unilamellar vesicles.PLoS One. 2011;6(10):e25529. doi: 10.1371/journal.pone.0025529. Epub 2011 Oct 6. PLoS One. 2011. PMID: 21998666 Free PMC article.
-
Electrophysiology in the eukaryotic model cell Saccharomyces cerevisiae.Pflugers Arch. 1998 Nov;436(6):999-1013. doi: 10.1007/s004240050735. Pflugers Arch. 1998. PMID: 9799419 Review.
-
Giant unilamellar vesicles - a perfect tool to visualize phase separation and lipid rafts in model systems.Acta Biochim Pol. 2009;56(1):33-9. Epub 2009 Mar 17. Acta Biochim Pol. 2009. PMID: 19287805 Review.
Cited by
-
Regulation of membrane protein structure and function by their lipid nano-environment.Nat Rev Mol Cell Biol. 2023 Feb;24(2):107-122. doi: 10.1038/s41580-022-00524-4. Epub 2022 Sep 2. Nat Rev Mol Cell Biol. 2023. PMID: 36056103 Free PMC article. Review.
-
Sterols in plant biology - Advances in studying membrane dynamics.Cell Surf. 2025 May 29;13:100147. doi: 10.1016/j.tcsw.2025.100147. eCollection 2025 Jun. Cell Surf. 2025. PMID: 40519718 Free PMC article. Review.
-
Dependence and Homeostasis of Membrane Impedance on Cell Morphology in Cultured Hippocampal Neurons.Sci Rep. 2018 Jul 2;8(1):9905. doi: 10.1038/s41598-018-28232-0. Sci Rep. 2018. PMID: 29967389 Free PMC article.
-
Cell-Derived Plasma Membrane Vesicles Are Permeable to Hydrophilic Macromolecules.Biophys J. 2020 Mar 24;118(6):1292-1300. doi: 10.1016/j.bpj.2019.12.040. Epub 2020 Jan 28. Biophys J. 2020. PMID: 32053777 Free PMC article.
-
[Study on the effect of small alcohol on lipid hydration and liposome formation].Sheng Wu Yi Xue Gong Cheng Xue Za Zhi. 2022 Feb 25;39(1):112-119. doi: 10.7507/1001-5515.202105060. Sheng Wu Yi Xue Gong Cheng Xue Za Zhi. 2022. PMID: 35231972 Free PMC article. Chinese.
References
-
- Hille B. Ion Channels of Excitable Membranes. 3rd Ed Sinauer; Sunderland, MA: 2001.
-
- Hamill OP, Marty A, Neher E, Sakmann B, Sigworth FJ. Improved patch-clamp techniques for high-resolution current recording from cells and cell-free membrane patches. Pflugers Arch. 1981;391(2):85–100. - PubMed
-
- Varghese A, Tenbroek EM, Coles J, Jr, Sigg DC. Endogenous channels in HEK cells and potential roles in HCN ionic current measurements. Prog Biophys Mol Biol. 2006;90(1-3):26–37. - PubMed
-
- Chandrasekhar KD, Bas T, Kobertz WR. KCNE1 subunits require co-assembly with K+ channels for efficient trafficking and cell surface expression. J Biol Chem. 2006;281(52):40015–40023. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

