Tensin 1 Is Essential for Myofibroblast Differentiation and Extracellular Matrix Formation
- PMID: 28005397
- PMCID: PMC5449508
- DOI: 10.1165/rcmb.2016-0104OC
Tensin 1 Is Essential for Myofibroblast Differentiation and Extracellular Matrix Formation
Abstract
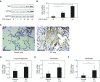
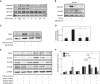
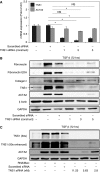
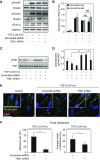
Myofibroblasts, the primary effector cells that mediate matrix remodeling during pulmonary fibrosis, rapidly assemble an extracellular fibronectin matrix. Tensin (TNS) 1 is a key component of specialized cellular adhesions (fibrillar adhesions) that bind to extracellular fibronectin fibrils. We hypothesized that TNS1 may play a role in modulating myofibroblast-mediated matrix formation. We found that TNS1 expression is increased in fibroblastic foci from lungs with idiopathic pulmonary fibrosis. Transforming growth factor (TGF)-β profoundly up-regulates TNS1 expression with kinetics that parallel the expression of the myofibroblast marker, smooth muscle α-actin. TGF-β-induced TNS1 expression is dependent on signaling through the TGF-β receptor 1 and is Rho coiled-coiled kinase/actin/megakaryoblastic leukemia-1/serum response factor dependent. Small interfering RNA-mediated knockdown of TNS1 disrupted TGF-β-induced myofibroblast differentiation, without affecting TGF-β/Smad signaling. In contrast, loss of TNS1 resulted in disruption of focal adhesion kinase phosphorylation, focal adhesion formation, and actin stress fiber development. Finally, TNS1 was essential for the formation of fibrillar adhesions and the assembly of nascent fibronectin and collagen matrix in myofibroblasts. In summary, our data show that TNS1 is a novel megakaryoblastic leukemia-1-dependent gene that is induced during pulmonary fibrosis. TNS1 plays an essential role in TGF-β-induced myofibroblast differentiation and myofibroblast-mediated formation of extracellular fibronectin and collagen matrix. Targeted disruption of TNS1 and associated signaling may provide an avenue to inhibit tissue fibrosis.
Keywords: extracellular matrix; fibrillar adhesion; megakaryoblastic leukemia-1; myofibroblast; tensin 1.
Figures

Similar articles
-
The Novel mTOR Complex 1/2 Inhibitor P529 Inhibits Human Lung Myofibroblast Differentiation.J Cell Biochem. 2017 Aug;118(8):2241-2249. doi: 10.1002/jcb.25878. Epub 2017 Apr 18. J Cell Biochem. 2017. PMID: 28078713 Free PMC article.
-
Delayed stress fiber formation mediates pulmonary myofibroblast differentiation in response to TGF-β.Am J Physiol Lung Cell Mol Physiol. 2011 Nov;301(5):L656-66. doi: 10.1152/ajplung.00166.2011. Epub 2011 Aug 19. Am J Physiol Lung Cell Mol Physiol. 2011. PMID: 21856814 Free PMC article.
-
Pharmacologic inhibition of lactate production prevents myofibroblast differentiation.Am J Physiol Lung Cell Mol Physiol. 2015 Dec 1;309(11):L1305-12. doi: 10.1152/ajplung.00058.2015. Epub 2015 Sep 25. Am J Physiol Lung Cell Mol Physiol. 2015. PMID: 26408551 Free PMC article.
-
Mechanisms of force generation and transmission by myofibroblasts.Curr Opin Biotechnol. 2003 Oct;14(5):538-46. doi: 10.1016/j.copbio.2003.08.006. Curr Opin Biotechnol. 2003. PMID: 14580586 Review.
-
Transforming growth factor-β signalling: role and consequences of Smad linker region phosphorylation.Cell Signal. 2013 Oct;25(10):2017-24. doi: 10.1016/j.cellsig.2013.06.001. Epub 2013 Jun 11. Cell Signal. 2013. PMID: 23770288 Review.
Cited by
-
Myeloid CD40 deficiency reduces atherosclerosis by impairing macrophages' transition into a pro-inflammatory state.Cardiovasc Res. 2023 May 22;119(5):1146-1160. doi: 10.1093/cvr/cvac084. Cardiovasc Res. 2023. PMID: 35587037 Free PMC article.
-
Contact Guidance of Connective Tissue Fibroblasts on Submicrometer Anisotropic Topographical Cues Is Dependent on Tissue of Origin, β1 Integrins, and Tensin-1 Recruitment.ACS Appl Mater Interfaces. 2023 Apr 26;15(16):19817-19832. doi: 10.1021/acsami.2c22381. Epub 2023 Apr 17. ACS Appl Mater Interfaces. 2023. PMID: 37067372 Free PMC article.
-
Bibliometric analysis of MXRA7 gene research trajectory: trends and insights (2015-2024).Discov Oncol. 2025 Jun 3;16(1):989. doi: 10.1007/s12672-025-02824-5. Discov Oncol. 2025. PMID: 40459788 Free PMC article.
-
ROCK Inhibition as Potential Target for Treatment of Pulmonary Hypertension.Cells. 2021 Jun 30;10(7):1648. doi: 10.3390/cells10071648. Cells. 2021. PMID: 34209333 Free PMC article. Review.
-
Effects of Shear Stress on Production of FVIII and vWF in a Cell-Based Therapeutic for Hemophilia A.Front Bioeng Biotechnol. 2021 Mar 1;9:639070. doi: 10.3389/fbioe.2021.639070. eCollection 2021. Front Bioeng Biotechnol. 2021. PMID: 33732691 Free PMC article.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

