Movement Initiation Signals in Mouse Whisker Motor Cortex
- PMID: 28009277
- PMCID: PMC5196025
- DOI: 10.1016/j.neuron.2016.12.001
Movement Initiation Signals in Mouse Whisker Motor Cortex
Abstract
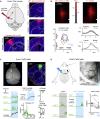
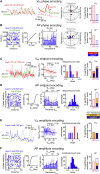
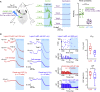
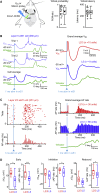
Frontal cortex plays a central role in the control of voluntary movements, which are typically guided by sensory input. Here, we investigate the function of mouse whisker primary motor cortex (wM1), a frontal region defined by dense innervation from whisker primary somatosensory cortex (wS1). Optogenetic stimulation of wM1 evokes rhythmic whisker protraction (whisking), whereas optogenetic inactivation of wM1 suppresses initiation of whisking. Whole-cell membrane potential recordings and silicon probe recordings of action potentials reveal layer-specific neuronal activity in wM1 at movement initiation, and encoding of fast and slow parameters of movements during whisking. Interestingly, optogenetic inactivation of wS1 caused hyperpolarization and reduced firing in wM1, together with reduced whisking. Optogenetic stimulation of wS1 drove activity in wM1 with complex dynamics, as well as evoking long-latency, wM1-dependent whisking. Our results advance understanding of a well-defined frontal region and point to an important role for sensory input in controlling motor cortex.
Keywords: action potential; membrane potential; motor coding; motor cortex; movement initiation; multisite silicon probe recording; optogenetics; sensorimotor integration; whisker motor control; whole-cell recording.
Copyright © 2016 The Authors. Published by Elsevier Inc. All rights reserved.
Figures

Similar articles
-
Sensorimotor processing in the rodent barrel cortex.Nat Rev Neurosci. 2019 Sep;20(9):533-546. doi: 10.1038/s41583-019-0200-y. Epub 2019 Jul 31. Nat Rev Neurosci. 2019. PMID: 31367018 Free PMC article. Review.
-
Parallel pathways from motor and somatosensory cortex for controlling whisker movements in mice.Eur J Neurosci. 2015 Feb;41(3):354-67. doi: 10.1111/ejn.12800. Epub 2014 Dec 5. Eur J Neurosci. 2015. PMID: 25476605 Free PMC article.
-
Interareal Synaptic Inputs Underlying Whisking-Related Activity in the Primary Somatosensory Barrel Cortex.J Neurosci. 2024 Jan 24;44(4):e1148232023. doi: 10.1523/JNEUROSCI.1148-23.2023. J Neurosci. 2024. PMID: 38050130 Free PMC article.
-
Optogenetic stimulation of cortex to map evoked whisker movements in awake head-restrained mice.Neuroscience. 2018 Jan 1;368:199-213. doi: 10.1016/j.neuroscience.2017.04.004. Epub 2017 Apr 12. Neuroscience. 2018. PMID: 28412497 Free PMC article.
-
Cortical control of whisker movement.Annu Rev Neurosci. 2014;37:183-203. doi: 10.1146/annurev-neuro-062012-170344. Epub 2014 May 9. Annu Rev Neurosci. 2014. PMID: 24821429 Review.
Cited by
-
Axonal connections between S1 barrel, M1, and S2 cortex in the newborn mouse.Front Neuroanat. 2023 Jan 25;17:1105998. doi: 10.3389/fnana.2023.1105998. eCollection 2023. Front Neuroanat. 2023. PMID: 36760662 Free PMC article.
-
Differential recordings of local field potential: A genuine tool to quantify functional connectivity.PLoS One. 2018 Dec 26;13(12):e0209001. doi: 10.1371/journal.pone.0209001. eCollection 2018. PLoS One. 2018. PMID: 30586445 Free PMC article.
-
The Cortical States of Wakefulness.Front Syst Neurosci. 2019 Jan 8;12:64. doi: 10.3389/fnsys.2018.00064. eCollection 2018. Front Syst Neurosci. 2019. PMID: 30670952 Free PMC article. Review.
-
Active information maintenance in working memory by a sensory cortex.Elife. 2019 Jun 24;8:e43191. doi: 10.7554/eLife.43191. Elife. 2019. PMID: 31232695 Free PMC article.
-
Sensorimotor processing in the rodent barrel cortex.Nat Rev Neurosci. 2019 Sep;20(9):533-546. doi: 10.1038/s41583-019-0200-y. Epub 2019 Jul 31. Nat Rev Neurosci. 2019. PMID: 31367018 Free PMC article. Review.
References
-
- Aronoff R., Matyas F., Mateo C., Ciron C., Schneider B., Petersen C.C.H. Long-range connectivity of mouse primary somatosensory barrel cortex. Eur. J. Neurosci. 2010;31:2221–2233. - PubMed
-
- Brecht M., Krauss A., Muhammad S., Sinai-Esfahani L., Bellanca S., Margrie T.W. Organization of rat vibrissa motor cortex and adjacent areas according to cytoarchitectonics, microstimulation, and intracellular stimulation of identified cells. J. Comp. Neurol. 2004;479:360–373. - PubMed
-
- Crochet S., Petersen C.C.H. Correlating whisker behavior with membrane potential in barrel cortex of awake mice. Nat. Neurosci. 2006;9:608–610. - PubMed
-
- Crochet S., Poulet J.F.A., Kremer Y., Petersen C.C.H. Synaptic mechanisms underlying sparse coding of active touch. Neuron. 2011;69:1160–1175. - PubMed
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

