Synaptic Specializations Support Frequency-Independent Purkinje Cell Output from the Cerebellar Cortex
- PMID: 28009294
- PMCID: PMC5870134
- DOI: 10.1016/j.celrep.2016.11.081
Synaptic Specializations Support Frequency-Independent Purkinje Cell Output from the Cerebellar Cortex
Abstract
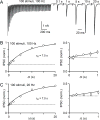
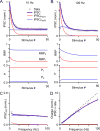
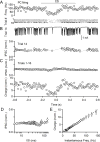
The output of the cerebellar cortex is conveyed to the deep cerebellar nuclei (DCN) by Purkinje cells (PCs). Here, we characterize the properties of the PC-DCN synapse in juvenile and adult mice and find that prolonged high-frequency stimulation leads to steady-state responses that become increasingly frequency independent within the physiological firing range of PCs in older animals, resulting in a linear relationship between charge transfer and activation frequency. We used a low-affinity antagonist to show that GABAA-receptor saturation occurs at this synapse but does not underlie frequency-invariant transmission. We propose that PC-DCN synapses have two components of release: one prominent early in trains and another specialized to maintain transmission during prolonged activation. Short-term facilitation offsets partial vesicle depletion to produce frequency-independent transmission.
Keywords: Purkinje cells; TPMPA; cerebellum; deep cerebellar nucleus; presynaptic; receptor saturation; recovery from depression; short-term facilitation.
Copyright © 2016 The Author(s). Published by Elsevier Inc. All rights reserved.
Figures




References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

