Metrological Timelines in Traceability
- PMID: 28009372
- PMCID: PMC4891962
- DOI: 10.6028/jres.103.005
Metrological Timelines in Traceability
Abstract
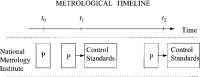
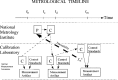
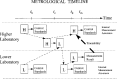
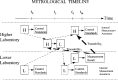
There is a growing requirement for an internationally accepted system of recognition of measurement capabilities and relationships within and among countries, to facilitate seamless global commerce and trade. As a result, metrologists worldwide have recently developed increased interest in the concept and definition of traceability. Classically, traceability provides a way of relating the results of a measurement (or value of a standard) to higher level standards. Such standards are usually national or international standards, and the comparisons used to provide the traceability must have well-understood uncertainties. An additional complexity arises because all instruments and standards are subject to change, however slight, over time. This paper develops approaches for dealing with the effects of such time-dependent changes as a part of traceability statements. The use of metrological time-lines provides a means of effectively visualizing these relationships in a statement of traceability. When the rate of change in the measurement process is sufficiently small, the approach proposed here is less important. However, documented measurement assurance procedures are required at all levels so that appropriate uncertainties may be estimated with confidence. When laboratory or national boundaries are crossed in the traceability process, other factors come into play, and the original concept of traceability can become obscure. It is becoming common to hear the term "equivalence" used to describe these more complex measurement relationships.
Keywords: calibration; equivalence; measurement assurance; measurement comparisons; measurement uncertainty; metrology; timelines; traceability; traceability statements.
Figures





References
-
- International Vocabulary of Basic and General Terms in Metrology. second edition. BIPM, IEC, IFCC, ISO, IUPAC, IUPAP, and OIML, International Organization for Standardization; Geneva: 1993. 1993.
-
- Quinn TJ. International Equivalence of National Measurement Standards; Note for Discussion at the Conference on International Equivalence of National Measurement Standards; Paris. 1997.
-
- Belanger BC. The Role of NBS Calibrations in Quality Assurance; ASIC Quality Congress Transactions; Boston. 1983.
-
- Rasberry SD. The Role of NBS SRMs in Quality Assurance; ASQC Quality Congress Transactions; Boston. 1983.
-
- Draft International Standard ISO/DIS 10012-2, Annex A. International Organization for Standardization; Geneva: 1996.
LinkOut - more resources
Full Text Sources
Research Materials
