Pharmacological characterization of N-[(2S)-5-(6-fluoro-3-pyridinyl)-2, 3-dihydro-1H-inden-2-yl]-2-propanesulfonamide: a novel, clinical AMPA receptor positive allosteric modulator
- PMID: 28009436
- PMCID: PMC5301047
- DOI: 10.1111/bph.13696
Pharmacological characterization of N-[(2S)-5-(6-fluoro-3-pyridinyl)-2, 3-dihydro-1H-inden-2-yl]-2-propanesulfonamide: a novel, clinical AMPA receptor positive allosteric modulator
Abstract
Background and purpose: AMPA receptor positive allosteric modulators represent a potential therapeutic strategy to improve cognition in people with schizophrenia. These studies collectively constitute the preclinical pharmacology data package used to build confidence in the pharmacology of this molecule and enable a clinical trial application.
Experimental approach: [N-[(2S)-5-(6-fluoro-3-pyridinyl)-2,3-dihydro 1H-inden-2-yl]-2-propanesulfonamide] (UoS12258) was profiled in a number of in vitro and in vivo studies to highlight its suitability as a novel therapeutic agent.
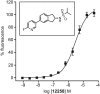
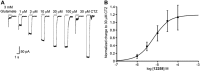
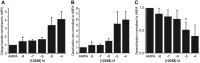
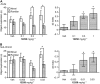
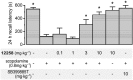
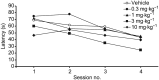
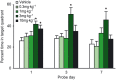
Key results: We demonstrated that UoS12258 is a selective, positive allosteric modulator of the AMPA receptor. At rat native hetero-oligomeric AMPA receptors, UoS12258 displayed a minimum effective concentration of approximately 10 nM in vitro and enhanced AMPA receptor-mediated synaptic transmission at an estimated free brain concentration of approximately 15 nM in vivo. UoS12258 reversed a delay-induced deficit in novel object recognition in rats after both acute and sub-chronic dosing. Sub-chronic dosing reduced the minimum effective dose from 0.3 to 0.03 mg·kg-1 . UoS12258 was also effective at improving performance in two other cognition models, passive avoidance in scopolamine-impaired rats and water maze learning and retention in aged rats. In side-effect profiling studies, UoS12258 did not produce significant changes in the maximal electroshock threshold test at doses below 10 mg·kg-1 .
Conclusion and implications: We conclude that UoS12258 is a potent and selective AMPA receptor modulator exhibiting cognition enhancing properties in several rat behavioural models superior to other molecules that have previously entered clinical evaluation.
© 2016 The British Pharmacological Society.
Figures


Similar articles
-
Pharmacological characterisation of MDI-222, a novel AMPA receptor positive allosteric modulator with an improved safety profile.J Psychopharmacol. 2020 Jan;34(1):93-102. doi: 10.1177/0269881119872198. Epub 2019 Nov 26. J Psychopharmacol. 2020. PMID: 31766938 Free PMC article.
-
Discovery of N-[(2S)-5-(6-fluoro-3-pyridinyl)-2,3-dihydro-1H-inden-2-yl]-2-propanesulfonamide, a novel clinical AMPA receptor positive modulator.J Med Chem. 2010 Aug 12;53(15):5801-12. doi: 10.1021/jm1005429. J Med Chem. 2010. PMID: 20614889 Clinical Trial.
-
7-Chloro-3-methyl-3-4-dihydro-2H-1,2,4 benzothiadiazine S,S-dioxide (IDRA 21): a benzothiadiazine derivative that enhances cognition by attenuating DL-alpha-amino-2,3-dihydro-5-methyl-3-oxo-4-isoxazolepropanoic acid (AMPA) receptor desensitization.J Pharmacol Exp Ther. 1995 Jan;272(1):300-9. J Pharmacol Exp Ther. 1995. PMID: 7815345
-
LY404187: a novel positive allosteric modulator of AMPA receptors.CNS Drug Rev. 2002 Fall;8(3):255-82. doi: 10.1111/j.1527-3458.2002.tb00228.x. CNS Drug Rev. 2002. PMID: 12353058 Free PMC article. Review.
-
Pharmacology of AMPA/kainate receptor ligands and their therapeutic potential in neurological and psychiatric disorders.Drugs. 2000 Jan;59(1):33-78. doi: 10.2165/00003495-200059010-00004. Drugs. 2000. PMID: 10718099 Review.
Cited by
-
Positron Emission Tomography (PET) Ligand Development for Ionotropic Glutamate Receptors: Challenges and Opportunities for Radiotracer Targeting N-Methyl-d-aspartate (NMDA), α-Amino-3-hydroxy-5-methyl-4-isoxazolepropionic Acid (AMPA), and Kainate Receptors.J Med Chem. 2019 Jan 24;62(2):403-419. doi: 10.1021/acs.jmedchem.8b00714. Epub 2018 Aug 27. J Med Chem. 2019. PMID: 30110164 Free PMC article. Review.
-
Schizophrenia: synthetic strategies and recent advances in drug design.Medchemcomm. 2018 Mar 16;9(5):759-782. doi: 10.1039/c7md00448f. eCollection 2018 May 1. Medchemcomm. 2018. PMID: 30108966 Free PMC article. Review.
-
Pharmacological characterisation of MDI-222, a novel AMPA receptor positive allosteric modulator with an improved safety profile.J Psychopharmacol. 2020 Jan;34(1):93-102. doi: 10.1177/0269881119872198. Epub 2019 Nov 26. J Psychopharmacol. 2020. PMID: 31766938 Free PMC article.
-
Positive AMPA and Kainate Receptor Modulators and Their Therapeutic Potential in CNS Diseases: A Comprehensive Review.Int J Mol Sci. 2025 Jul 4;26(13):6450. doi: 10.3390/ijms26136450. Int J Mol Sci. 2025. PMID: 40650226 Free PMC article. Review.
-
Radiosynthesis and preliminary evaluation of 11C-labeled 4-cyclopropyl-7-(3-methoxyphenoxy)-3,4-dihydro-2H-benzo[e] [1,2,4] thiadiazine 1,1-dioxide for PET imaging AMPA receptors.Tetrahedron Lett. 2020 Mar 19;61(12):151635. doi: 10.1016/j.tetlet.2020.151635. Epub 2020 Jan 17. Tetrahedron Lett. 2020. PMID: 32153306 Free PMC article.
References
-
- Akbarian S, Smith MA, Jones EG (1995). Editing for an AMPA receptor subunit RNA in prefrontal cortex and striatum in Alzheimer's disease, Huntington's disease and schizophrenia. Brain Res 699: 297–304. - PubMed
-
- Arai A, Kessler M, Ambros‐Ingerson J, Quan A, Yigiter E, Rogers G et al. (1996). Effects of a centrally active benzoylpyrrolidine drug on AMPA receptor kinetics. Neuroscience 75: 573–585. - PubMed
-
- Beneyto M, Meador‐Woodruff JH (2004). Expression of transcripts encoding AMPA receptor subunits and associated postsynaptic proteins in the macaque brain. J Comp Neurol 468: 530–554. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

