Induction of H7N9-Cross-Reactive Antibody-Dependent Cellular Cytotoxicity Antibodies by Human Seasonal Influenza A Viruses that are Directed Toward the Nucleoprotein
- PMID: 28011910
- PMCID: PMC5853654
- DOI: 10.1093/infdis/jiw629
Induction of H7N9-Cross-Reactive Antibody-Dependent Cellular Cytotoxicity Antibodies by Human Seasonal Influenza A Viruses that are Directed Toward the Nucleoprotein
Abstract
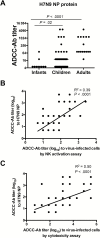
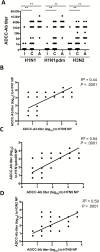
Antibodies that mediate antibody-dependent cellular cytotoxicity (ADCC) against avian influenza virus subtypes, including H7N9 and H5N1, have been detected in human sera. Using NK cell activation and NK cytotoxicity assays, we compared ADCC-mediating antibodies (ADCC-Abs) in sera collected from healthy infants, children and adults against H7N9 virus-infected cells and recombinant hemagglutinin (HA), neuraminidase (NA), and nucleoprotein (NP) proteins. High titers of ADCC-Abs against H7N9 virus-infected cells were detected in sera from adults and children but not infants. ADCC-Abs titers directed against H7N9 HA or NA proteins. Further analysis showed that ADCC-Abs titers were significantly higher toward H7N9 NP, as compared with H7N9 HA or NA proteins, and correlated strongly with ADCC-Abs titers against H7N9 virus-infected cells. Indeed, ADCC-Abs to NPs of seasonal H1N1 and H3N2 viruses correlated strongly with ADCC-Abs to H7N9 NP, suggesting that seasonal influenza infections and vaccinations may induce these cross-reactive antibodies. Targeting ADCC-Abs to internal proteins may be a potential mechanism of universal vaccine design.
Keywords: ADCC; antibody; antibody-dependent cellular cytotoxicity; influenza virus; nucleoprotein.
© The Author 2016. Published by Oxford University Press for the Infectious Diseases Society of America. All rights reserved. For permissions, e-mail journals.permissions@oup.com.
Figures
References
-
- World Health Organization. WHO risk assessment of human infections with avian influenza A(H7N9) virus. http://www.who.int/influenza/human_animal_interface/influenza_h7n9/RiskA... Accessed 27 Septemeber 2016.
-
- Jackson LA, Campbell JD, Frey SE, et al. Effect of varying doses of a monovalent H7N9 influenza vaccine with and without AS03 and MF59 adjuvants on immune response: a randomized clinical trial. JAMA 2015; 314:237–46. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

