The intersection of radiotherapy and immunotherapy: mechanisms and clinical implications
- PMID: 28018989
- PMCID: PMC5171206
- DOI: 10.1126/sciimmunol.aag1266
The intersection of radiotherapy and immunotherapy: mechanisms and clinical implications
Abstract
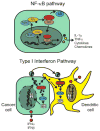
By inducing DNA damage, radiotherapy both reduces tumor burden and enhances anti-tumor immunity. Here, we will review the mechanisms by which radiation induces anti-tumor immune responses that can be augmented using immunotherapies to facilitate tumor regression. Radiotherapy increases inflammation in tumors by activating the NF-κB and the Type I interferon response pathways to induce expression of pro-inflammatory cytokines. This inflammation coupled with antigen release from irradiated cells facilitates dendritic cell maturation and cross-presentation of tumor antigens to prime tumor-specific T cell responses. Radiation also sensitizes tumors to these T cell responses by enhancing T cell infiltration into tumors and the recognition of both malignant cancer cells and non-malignant stroma that present cognate antigen. Yet, these anti-tumor immune responses may be blunted by several mechanisms including regulatory T cells and checkpoint molecules that promote T cell tolerance and exhaustion. Consequently, the combination of immunotherapy using vaccines and/or checkpoint inhibitors with radiation is demonstrating early clinical potential. Overall, this review will provide a global view for how radiation and the immune system converge to target cancers and the early attempts to exploit this synergy in clinical practice.
Keywords: Checkpoint blockade; Immunotherapy; Radiotherapy.
Figures



References
-
- Stone HB, Peters LJ, Milas L. Effect of host immune capability on radiocurability and subsequent transplantability of a murine fibrosarcoma. J Natl Cancer Inst. 1979;63:1229–1235. - PubMed
-
- Lugade AA, Sorensen EW, Gerber SA, Moran JP, Frelinger JG, Lord EM. Radiation-induced IFN-gamma production within the tumor microenvironment influences antitumor immunity. Journal of immunology. 2008;180:3132–3139. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical