A novel Chk1-binding peptide that enhances genotoxic sensitivity through the cellular redistribution of nuclear Chk1
- PMID: 28025997
- PMCID: PMC5065296
- DOI: 10.3892/ijmm.2016.2762
A novel Chk1-binding peptide that enhances genotoxic sensitivity through the cellular redistribution of nuclear Chk1
Abstract
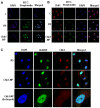
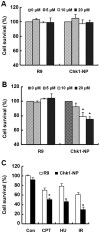
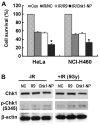
Since checkpoint kinase 1 (Chk1) is an essential factor for cell viability following DNA damage, the inhibition of Chk1 has been a major focus of pharmaceutical development to enhance the sensitivity of tumor cells to chemo- and radiotherapy that damage DNA. However, due to the off-target effects of conventional Chk1-targeting strategies and the toxicity of Chk1 inhibitors, alternative strategies are required to target Chk1. To facilitate such efforts, in this study, we identified a specific Chk1-binding 12-mer peptide from the screening of a phage display library and characterized the peptide in terms of cellular cytotoxicity, and in terms of its effect on Chk1 activity and sensitivity to genotoxic agents. This peptide, named N-terminal Chk1-binding peptide (Chk1‑NP), bound the kinase domain of Chk1. Simulation of the binding revealed that the very N-terminus of the Chk1 kinase domain is the potential peptide binding site. Of note, the polyarginine-mediated internalization of Chk1‑NP redistributed nuclear Chk1 with a prominent decrease in the nucleus in the absence of DNA damage. Treatment with Chk1‑NP peptide alone decreased the viability of p53-defective HeLa cells, but not that of p53-functional NCI-H460 cells under normal conditions. The treatment of HeLa or NCI-H460 cells with the peptide significantly enhanced radiation sensitivity following ionizing radiation (IR) with a greater enhancement observed in HeLa cells. Moreover, the IR-induced destabilization of Chk1 was aggravated by treatment with Chk1‑NP. Therefore, the decreased nuclear localization and protein levels of Chk1 seem to be responsible for the enhanced cancer cell killing following combined treatment with IR and Chk1‑NP. The approach using the specific Chk1-binding peptide may facilitate the mechanistic understanding and potential modulation of Chk1 activities and may provide a novel rationale for the development of specific Chk1-targeting agents.
Figures



Similar articles
-
Enhancement of cellular radiation sensitivity through degradation of Chk1 by the XIAP-XAF1 complex.Cancer Biol Ther. 2014;15(12):1622-34. doi: 10.4161/15384047.2014.962305. Cancer Biol Ther. 2014. PMID: 25535897 Free PMC article.
-
A novel mechanism of checkpoint abrogation conferred by Chk1 downregulation.Oncogene. 2005 Feb 17;24(8):1403-11. doi: 10.1038/sj.onc.1208309. Oncogene. 2005. PMID: 15608676
-
A small peptide mimicking the key domain of MEPE/OF45 interacting with CHK1 protects human cells from radiation-induced killing.Cell Cycle. 2010 May 15;9(10):1981-5. doi: 10.4161/cc.9.10.11651. Epub 2010 May 15. Cell Cycle. 2010. PMID: 20436300 Free PMC article.
-
Interferon-gamma enhances radiation-induced cell death via downregulation of Chk1.Cancer Biol Ther. 2012 Sep;13(11):1018-25. doi: 10.4161/cbt.20990. Epub 2012 Jul 24. Cancer Biol Ther. 2012. PMID: 22825336 Free PMC article.
-
The ATM-Chk2 and ATR-Chk1 pathways in DNA damage signaling and cancer.Adv Cancer Res. 2010;108:73-112. doi: 10.1016/B978-0-12-380888-2.00003-0. Adv Cancer Res. 2010. PMID: 21034966 Review.
References
-
- Sørensen CS, Syljuåsen RG, Falck J, Schroeder T, Rönnstrand L, Khanna KK, Zhou BB, Bartek J, Lukas J. Chk1 regulates the S phase checkpoint by coupling the physiological turnover and ionizing radiation-induced accelerated proteolysis of Cdc25A. Cancer Cell. 2003;3:247–258. doi: 10.1016/S1535-6108(03)00048-5. - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

