Pharmacokinetics/Pharmacodynamics of Pulmonary Delivery of Colistin against Pseudomonas aeruginosa in a Mouse Lung Infection Model
- PMID: 28031207
- PMCID: PMC5328518
- DOI: 10.1128/AAC.02025-16
Pharmacokinetics/Pharmacodynamics of Pulmonary Delivery of Colistin against Pseudomonas aeruginosa in a Mouse Lung Infection Model
Abstract
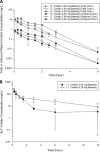
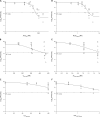
Colistin is often administered by inhalation and/or the parenteral route for the treatment of respiratory infections caused by multidrug-resistant (MDR) Pseudomonas aeruginosa However, limited pharmacokinetic (PK) and pharmacodynamic (PD) data are available to guide the optimization of dosage regimens of inhaled colistin. In the present study, PK of colistin in epithelial lining fluid (ELF) and plasma was determined following intratracheal delivery of a single dose of colistin solution in neutropenic lung-infected mice. The antimicrobial efficacy of intratracheal delivery of colistin against three P. aeruginosa strains (ATCC 27853, PAO1, and FADDI-PA022; MIC of 1 mg/liter for all strains) was examined in a neutropenic mouse lung infection model. Dose fractionation studies were conducted over 2.64 to 23.8 mg/kg of body weight/day. The inhibitory sigmoid model was employed to determine the PK/PD index that best described the antimicrobial efficacy of pulmonary delivery of colistin. In both ELF and plasma, the ratio of the area under the unbound concentration-time profile to MIC (fAUC/MIC) was the PK/PD index that best described the antimicrobial effect in mouse lung infection (R2 = 0.60 to 0.84 for ELF and 0.64 to 0.83 for plasma). The fAUC/MIC targets required to achieve stasis against the three strains were 684 to 1,050 in ELF and 2.15 to 3.29 in plasma. The histopathological data showed that pulmonary delivery of colistin reduced infection-caused pulmonary inflammation and preserved the integrity of the lung epithelium, although colistin introduced mild pulmonary inflammation in healthy mice. This study showed pulmonary delivery of colistin provides antimicrobial effects against MDR P. aeruginosa lung infections superior to those of parenteral administrations. For the first time, our results provide important preclinical PK/PD information for optimization of inhaled colistin therapy.
Keywords: Pseudomonas aeruginosa; colistin; intratracheal administration; mouse lung infection model; multidrug-resistant bacteria; pharmacodynamics; pharmacokinetics; respiratory tract infection.
Copyright © 2017 American Society for Microbiology.
Figures

Similar articles
-
Aerosolized Polymyxin B for Treatment of Respiratory Tract Infections: Determination of Pharmacokinetic-Pharmacodynamic Indices for Aerosolized Polymyxin B against Pseudomonas aeruginosa in a Mouse Lung Infection Model.Antimicrob Agents Chemother. 2017 Jul 25;61(8):e00211-17. doi: 10.1128/AAC.00211-17. Print 2017 Aug. Antimicrob Agents Chemother. 2017. PMID: 28559256 Free PMC article.
-
Elucidation of the pharmacokinetic/pharmacodynamic determinant of colistin activity against Pseudomonas aeruginosa in murine thigh and lung infection models.Antimicrob Agents Chemother. 2010 Mar;54(3):1117-24. doi: 10.1128/AAC.01114-09. Epub 2009 Dec 22. Antimicrob Agents Chemother. 2010. PMID: 20028824 Free PMC article.
-
New pharmacokinetic/pharmacodynamic studies of systemically administered colistin against Pseudomonas aeruginosa and Acinetobacter baumannii in mouse thigh and lung infection models: smaller response in lung infection.J Antimicrob Chemother. 2015 Dec;70(12):3291-7. doi: 10.1093/jac/dkv267. Epub 2015 Aug 27. J Antimicrob Chemother. 2015. PMID: 26318190
-
In vitro Pharmacodynamics and PK/PD in Animals.Adv Exp Med Biol. 2019;1145:105-116. doi: 10.1007/978-3-030-16373-0_8. Adv Exp Med Biol. 2019. PMID: 31364074 Review.
-
The clinical use of colistin in patients with cystic fibrosis.Curr Opin Pulm Med. 2001 Nov;7(6):434-40. doi: 10.1097/00063198-200111000-00013. Curr Opin Pulm Med. 2001. PMID: 11706322 Review.
Cited by
-
Model-Based Drug Development in Pulmonary Delivery: Pharmacokinetic Analysis of Novel Drug Candidates for Treatment of Pseudomonas aeruginosa Lung Infection.J Pharm Sci. 2019 Jan;108(1):630-640. doi: 10.1016/j.xphs.2018.09.017. Epub 2018 Sep 23. J Pharm Sci. 2019. PMID: 30257195 Free PMC article.
-
Application of the Adverse Outcome Pathway Concept to In Vitro Nephrotoxicity Assessment: Kidney Injury due to Receptor-Mediated Endocytosis and Lysosomal Overload as a Case Study.Front Toxicol. 2022 Apr 19;4:864441. doi: 10.3389/ftox.2022.864441. eCollection 2022. Front Toxicol. 2022. PMID: 35516525 Free PMC article.
-
Correlations between surface composition and aerosolization of jet-milled dry powder inhaler formulations with pharmaceutical lubricants.Int J Pharm. 2019 Sep 10;568:118504. doi: 10.1016/j.ijpharm.2019.118504. Epub 2019 Jul 9. Int J Pharm. 2019. PMID: 31299339 Free PMC article.
-
Composite particle formulations of colistin and meropenem with improved in-vitro bacterial killing and aerosolization for inhalation.Int J Pharm. 2018 Sep 5;548(1):443-453. doi: 10.1016/j.ijpharm.2018.07.010. Epub 2018 Jul 3. Int J Pharm. 2018. PMID: 30008433 Free PMC article.
-
Proof-of-Principle Study in a Murine Lung Infection Model of Antipseudomonal Activity of Phage PEV20 in a Dry-Powder Formulation.Antimicrob Agents Chemother. 2018 Jan 25;62(2):e01714-17. doi: 10.1128/AAC.01714-17. Print 2018 Feb. Antimicrob Agents Chemother. 2018. PMID: 29158280 Free PMC article.
References
-
- Mohamed AF, Cars O, Friberg LE. 2014. A pharmacokinetic/pharmacodynamic model developed for the effect of colistin on Pseudomonas aeruginosa in vitro with evaluation of population pharmacokinetic variability on simulated bacterial killing. J Antimicrob Chemother 69:1350–1361. doi:10.1093/jac/dkt520. - DOI - PubMed
-
- Cheah S-E, Wang J, Turnidge JD, Li J, Nation RL. 2015. New pharmacokinetic/pharmacodynamic studies of systemically administered colistin against Pseudomonas aeruginosa and Acinetobacter baumannii in mouse thigh and lung infection models: smaller response in lung infection. J Antimicrob Chemother 70:3291–3297. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

