Pinoresinol Diglucoside Alleviates oxLDL-Induced Dysfunction in Human Umbilical Vein Endothelial Cells
- PMID: 28042303
- PMCID: PMC5155123
- DOI: 10.1155/2016/3124519
Pinoresinol Diglucoside Alleviates oxLDL-Induced Dysfunction in Human Umbilical Vein Endothelial Cells
Abstract
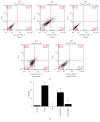
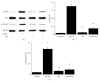
Atherosclerotic cardiovascular diseases are the leading causes of morbidity and mortality worldwide. Deposition of oxidized low-density lipoprotein (oxLDL) is one of the initiators and promoters of atherosclerosis. Eucommia lignans were shown to possess antihypertensive effects. This study aimed to investigate the effects of pinoresinol diglucoside (PD), a Eucommia lignan, on oxLDL-induced endothelial dysfunction. HUVECs were treated with oxLDL and/or PD followed by assessing radical oxygen species (ROS), apoptosis, nitrogen oxide (NO), malondialdehyde (MDA), and superoxide dismutase (SOD) activity with specific assays kits, mRNA levels with quantitative real-time polymerase chain reaction (PCR), and protein levels with western blot. PD abolished oxLDL-induced ROS and MDA production, apoptosis, upregulation of lectin-like oxidized LDL recptor-1 (LOX-1), intercellular Adhesion Molecule 1 (ICAM-1), and nuclear factor kappa-light-chain-enhancer of activated B-cells (NF-κB), and activation of p38MAPK (mitogen-activated protein kinases)/NF-κB signaling. Meanwhile, PD alleviated oxLDL-caused inhibition of SOD activity, eNOS expression, and NO production. These data demonstrated that PD was effective in protecting endothelial cells from oxLDL-caused injuries, which guarantees further investigation on the clinical benefits of PD on cardiovascular diseases.
Conflict of interest statement
All authors declare that they have no conflict of interests.
Figures





References
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

