Hippocampal encoding of interoceptive context during fear conditioning
- PMID: 28045462
- PMCID: PMC5545722
- DOI: 10.1038/tp.2016.254
Hippocampal encoding of interoceptive context during fear conditioning
Abstract
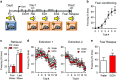
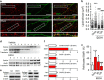
Rodent models of auditory fear conditioning are often used to understand the molecular mechanisms regulating fear- and anxiety-related behaviors. Conditioning and extinction memories are influenced by contextual cues, and the reinstatement of conditioned fear occurs when the conditioning stimulus is presented in a context different from the extinction context. Although it has been proposed that internal state is a feature of context that could influence extinction, contributions of interoception to conditioning have not been experimentally addressed. Here we use ethanol (EtOH) to show that interoceptive cues are encoded through the hippocampus by mechanisms that involve increased phosphorylation of GluR1 on serine 845, and biophysical alterations in neuronal membranes that facilitate stabilization of surface-located calcium-permeable n-2-amino-3-(5-methyl-3-oxo-1,2-oxazol-4-yl) propanoic acid (AMPA) receptor (AMPAR) into membrane microdomains. Conflicting interoceptive cues during extinction and fear relapse testing resulted in a failure to consolidate extinction that was reversed by the administration of AMPAR antagonists immediately following the retrieval cue.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- Lee JL. Memory reconsolidation mediates the strengthening of memories by additional learning. Nat Neurosci 2008; 11: 1264–1266. - PubMed
-
- Lee S, Song B, Kim J, Park K, Hong I, An B et al. GluA1 phosphorylation at serine 831 in the lateral amygdala is required for fear renewal. Nat Neurosci 2013; 16: 1436–1444. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

