Spectroscopic Studies of the EutT Adenosyltransferase from Salmonella enterica: Evidence of a Tetrahedrally Coordinated Divalent Transition Metal Cofactor with Cysteine Ligation
- PMID: 28045498
- PMCID: PMC5241240
- DOI: 10.1021/acs.biochem.6b00750
Spectroscopic Studies of the EutT Adenosyltransferase from Salmonella enterica: Evidence of a Tetrahedrally Coordinated Divalent Transition Metal Cofactor with Cysteine Ligation
Abstract
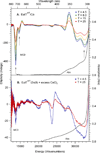
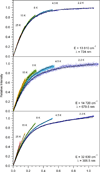
The EutT enzyme from Salmonella enterica, a member of the family of ATP:cobalt(I) corrinoid adenosyltransferase (ACAT) enzymes, requires a divalent transition metal ion for catalysis, with Fe(II) yielding the highest activity. EutT contains a unique cysteine-rich HX11CCX2C(83) motif (where H and the last C occupy the 67th and 83rd positions, respectively, in the amino acid sequence) not found in other ACATs and employs an unprecedented mechanism for the formation of adenosylcobalamin. Recent kinetic and spectroscopic studies of this enzyme revealed that residues in the HX11CCX2C(83) motif are required for the tight binding of the divalent metal ion and are critical for the formation of a four-coordinate (4c) cob(II)alamin [Co(II)Cbl] intermediate in the catalytic cycle. However, it remained unknown which, if any, of the residues in the HX11CCX2C(83) motif bind the divalent metal ion. To address this issue, we have characterized Co(II)-substituted wild-type EutT (EutTWT/Co) by using electronic absorption, electron paramagnetic resonance, and magnetic circular dichroism (MCD) spectroscopies. Our results indicate that the reduced catalytic activity of EutTWT/Co relative to that of the Fe(II)-containing enzyme arises from the incomplete incorporation of Co(II) ions and, thus, a decrease in the relative population of 4c Co(II)Cbl. Our MCD data for EutTWT/Co also reveal that the Co(II) ions reside in a distorted tetrahedral coordination environment with direct cysteine sulfur ligation. Additional spectroscopic studies of EutT/Co variants possessing a single alanine substitution of either His67, His75, Cys79, Cys80, or Cys83 indicate that Cys80 coordinates to the Co(II) ion, while the additional residues are important for maintaining the structural integrity and/or high affinity of the metal binding site.
Figures






Similar articles
-
Spectroscopic Study of the EutT Adenosyltransferase from Listeria monocytogenes: Evidence for the Formation of a Four-Coordinate Cob(II)alamin Intermediate.Biochemistry. 2018 Aug 28;57(34):5088-5095. doi: 10.1021/acs.biochem.8b00743. Epub 2018 Aug 16. Biochemistry. 2018. PMID: 30071158 Free PMC article.
-
Spectroscopic Studies of the EutT Adenosyltransferase from Salmonella enterica: Mechanism of Four-Coordinate Co(II)Cbl Formation.J Am Chem Soc. 2016 Mar 23;138(11):3694-704. doi: 10.1021/jacs.5b11708. Epub 2016 Mar 9. J Am Chem Soc. 2016. PMID: 26886077 Free PMC article.
-
the Eutt enzyme of Salmonella enterica is a unique ATP:Cob(I)alamin adenosyltransferase metalloprotein that requires ferrous ions for maximal activity.J Bacteriol. 2014 Feb;196(4):903-10. doi: 10.1128/JB.01304-13. Epub 2013 Dec 13. J Bacteriol. 2014. PMID: 24336938 Free PMC article.
-
Adenosyltransferase: an enzyme and an escort for coenzyme B12?Trends Biochem Sci. 2005 Jun;30(6):304-8. doi: 10.1016/j.tibs.2005.04.008. Trends Biochem Sci. 2005. PMID: 15950874 Review.
-
Multiple roles of ATP:cob(I)alamin adenosyltransferases in the conversion of B12 to coenzyme B12.Appl Microbiol Biotechnol. 2010 Sep;88(1):41-8. doi: 10.1007/s00253-010-2773-2. Epub 2010 Jul 31. Appl Microbiol Biotechnol. 2010. PMID: 20677021 Free PMC article. Review.
Cited by
-
Mutational and Functional Analyses of Substrate Binding and Catalysis of the Listeria monocytogenes EutT ATP:Co(I)rrinoid Adenosyltransferase.Biochemistry. 2020 Mar 17;59(10):1124-1136. doi: 10.1021/acs.biochem.0c00078. Epub 2020 Mar 9. Biochemistry. 2020. PMID: 32125848 Free PMC article.
-
A New Class of EutT ATP:Co(I)rrinoid Adenosyltransferases Found in Listeria monocytogenes and Other Firmicutes Does Not Require a Metal Ion for Activity.Biochemistry. 2018 Aug 28;57(34):5076-5087. doi: 10.1021/acs.biochem.8b00715. Epub 2018 Aug 16. Biochemistry. 2018. PMID: 30071718 Free PMC article.
-
Spectroscopic Study of the EutT Adenosyltransferase from Listeria monocytogenes: Evidence for the Formation of a Four-Coordinate Cob(II)alamin Intermediate.Biochemistry. 2018 Aug 28;57(34):5088-5095. doi: 10.1021/acs.biochem.8b00743. Epub 2018 Aug 16. Biochemistry. 2018. PMID: 30071158 Free PMC article.
References
-
- Warren MJ, Raux E, Schubert HL, Escalante-Semerena JC. The Biosynthesis of Adenosylcobalamin (vitamin B12) Nat. Prod. Rep. 2002;19(4):390–412. - PubMed
-
- Roth JR, Lawrence JG, Bobik TA. Cobalamin (coenzyme B12): Synthesis and Biological Significance. Annual review of microbiology. 1996:137–181. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

