Perfluoroalkyl acids-induced liver steatosis: Effects on genes controlling lipid homeostasis
- PMID: 28049043
- PMCID: PMC5994610
- DOI: 10.1016/j.tox.2016.12.007
Perfluoroalkyl acids-induced liver steatosis: Effects on genes controlling lipid homeostasis
Abstract
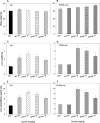
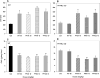
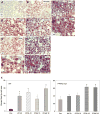
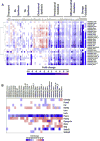
Persistent presence of perfluoroalkyl acids (PFAAs) in the environment is due to their extensive use in industrial and consumer products, and their slow decay. Biochemical tests in rodent demonstrated that these chemicals are potent modifiers of lipid metabolism and cause hepatocellular steatosis. However, the molecular mechanism of PFAAs interference with lipid metabolism remains to be elucidated. Currently, two major hypotheses are that PFAAs interfere with mitochondrial beta-oxidation of fatty acids and/or they affect the transcriptional activity of peroxisome proliferator-activated receptor α (PPARα) in liver. To determine the ability of structurally-diverse PFAAs to cause steatosis, as well as to understand the underlying molecular mechanisms, wild-type (WT) and PPARα-null mice were treated with perfluorooctanoic acid (PFOA), perfluorononanoic acid (PFNA), or perfluorohexane sulfonate (PFHxS), by oral gavage for 7days, and their effects were compared to that of PPARα agonist WY-14643 (WY), which does not cause steatosis. Increases in liver weight and cell size, and decreases in DNA content per mg of liver, were observed for all compounds in WT mice, and were also seen in PPARα-null mice for PFOA, PFNA, and PFHxS, but not for WY. In Oil Red O stained sections, WT liver showed increased lipid accumulation in all treatment groups, whereas in PPARα-null livers, accumulation was observed after PFNA and PFHxS treatment, adding to the burden of steatosis observed in control (untreated) PPARα-null mice. Liver triglyceride (TG) levels were elevated in WT mice by all PFAAs and in PPARα-null mice only by PFNA. In vitro β-oxidation of palmitoyl carnitine by isolated rat liver mitochondria was not inhibited by any of the 7 PFAAs tested. Likewise, neither PFOA nor PFOS inhibited palmitate oxidation by HepG2/C3A human liver cell cultures. Microarray analysis of livers from PFAAs-treated mice indicated that the PFAAs induce the expression of the lipid catabolism genes, as well as those involved in fatty acid and triglyceride synthesis, in WT mice and, to a lesser extent, in PPARα-null mice. These results indicate that most of the PFAAs increase liver TG load and promote steatosis in mice We hypothesize that PFAAs increase steatosis because the balance of fatty acid accumulation/synthesis and oxidation is disrupted to favor accumulation.
Keywords: Perfluorohexane sulfonate; Perfluorononanoic acid; Perfluorooctane sulfonate; Perfluorooctanoic acid; Steatosis; Triglycerides.
Published by Elsevier B.V.
Figures
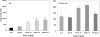


References
-
- Anderson SP, Dunn C, Laughter A, Yoon L, Swanson C, Stulnig TM, Steffensen KR, Chandraratna RA, Gustafsson JA, Corton JC. Overlapping transcriptional programs regulated by the nuclear receptors peroxisome proliferator-activated receptor alpha, retinoid X receptor, and liver X receptor in mouse liver. Mol Pharmacol. 2004;66:1440–1452. - PubMed
-
- Angrish M, Kaiser J, McQueen C, Chorley B. Tipping the balance: hepatotoxicity and the 4 apical key events of hepatic steatosis. Toxicol Sci. 2016;150:261–268. - PubMed
-
- Begriche K, Massart J, Robin MA, Bonnet F, Fromently B. Mitochondrial adaptations and dysfunctions in nonalcoholic fatty liver disease. Hepatology. 2013;58:1497–150. - PubMed
-
- Berthiaume J, Wallace KB. Perfluorooctanoate, perflourooctanesulfonate, and N-ethyl perfluorooctanesulfonamido ethanol; peroxisome proliferation and mitochondrial biogenesis. Toxicol Lett. 2002;129:23–32. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

