Targeting P-glycoprotein function, p53 and energy metabolism: Combination of metformin and 2-deoxyglucose reverses the multidrug resistance of MCF-7/Dox cells to doxorubicin
- PMID: 28052008
- PMCID: PMC5352427
- DOI: 10.18632/oncotarget.14373
Targeting P-glycoprotein function, p53 and energy metabolism: Combination of metformin and 2-deoxyglucose reverses the multidrug resistance of MCF-7/Dox cells to doxorubicin
Abstract
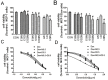
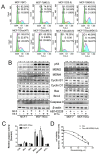
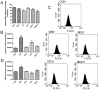
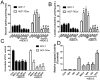
Multidrug resistance(MDR) is a major obstacle to efficiency of breast cancer chemotherapy. We investigated whether combination of metformin and 2-deoxyglucose reverses MDR of MCF-7/Dox cells and tried to elucidate the possible mechanisms. The combination of metformin and 2-deoxyglucose selectively enhanced cytotoxicity of doxorubicin against MCF-7/Dox cells. Combination of the two drugs resumed p53 function via inhibiting overexpression of murine doubleminute 2(MDM2) and murine doubleminute 4(MDM4) leading to G2/M arrest and apoptosis in MCF-7/Dox cells. Combination of the two drugs had no effect on P-glycoprotein mRNA expression and P-glycoprotein ATPase activity but increased doxorubicin accumulation in MCF-7/Dox cells. The increased doxorubicin accumulation maybe associate with metabolic stress. Combination of metformin and 2-deoxyglucose initiated a strong metabolic stress in MCF-7/Dox cells via inhibiting glucose uptake, lactate, fatty acid, ATP production and protein kinase B(AKT)/ mammalian target of rapamycin(mTOR) pathway. Taken together, combination of metformin and 2-deoxyglucose reverses MDR of MCF-7/Dox cells by recovering p53 function and increasing doxorubicin accumulation. Furthermore, doxorubicin selectively increases MCF-7/Dox apoptosis via aggravating metabolic stress induced by metformin plus 2-deoxyglucose. The mutually reinforcing effect made the combination of metformin and 2DG had a better effect on reversing MDR.
Keywords: P-glycoprotein; energy metabolism; multidrug resistance; p53.
Conflict of interest statement
The authors declare no financial or other conflict of interest with regard to this work.
Figures

References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

