Fgfr2 is integral for bladder mesenchyme patterning and function
- PMID: 28052872
- PMCID: PMC5407073
- DOI: 10.1152/ajprenal.00463.2016
Fgfr2 is integral for bladder mesenchyme patterning and function
Abstract
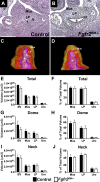
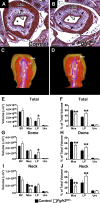
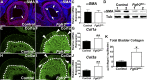
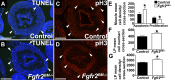
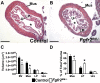
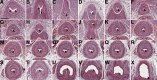
While urothelial signals, including sonic hedgehog (Shh), drive bladder mesenchyme differentiation, it is unclear which pathways within the mesenchyme are critical for its development. Studies have shown that fibroblast growth factor receptor 2 (Fgfr2) is necessary for kidney and ureter mesenchymal development. Our objective was to determine the role of Fgfr2 in bladder mesenchyme. We used Tbx18cre mice to delete Fgfr2 in bladder mesenchyme (Fgfr2BM-/-). We performed three-dimensional reconstructions, quantitative real-time PCR, in situ hybridization, immunolabeling, ELISAs, immunoblotting, void stain on paper, ex vivo bladder sheet assays, and in vivo decerebrated cystometry. Compared with controls, embryonic (E) day 16.5 (E16.5) Fgfr2BM-/- bladders have thin muscle layers with reduced α-smooth muscle actin levels and thickened lamina propria with increased collagen expression that intrudes into muscle. From postnatal (P) day 1 (P1) to P30, Fgfr2BM-/- bladders demonstrate progressive muscle loss and increased collagen expression. Postnatal Fgfr2BM-/- bladder sheets exhibit decreased contractility and increased passive stretch tension compared with controls. In vivo cystometry revealed high baseline and threshold pressures and shortened intercontractile intervals in Fgfr2BM-/- bladders compared with controls. Mechanistically, while Shh expression appears normal, mRNA and protein readouts of hedgehog activity are increased in E16.5 Fgfr2BM-/- bladders compared with controls. Moreover, E16.5Fgfr2BM-/- bladders exhibit higher levels of Cdo and Boc, hedgehog coreceptors that enhance sensitivity to Shh, than controls. Fgfr2 is critical for bladder mesenchyme patterning by virtue of its role in modulation of hedgehog signaling.
Keywords: bladder development; bladder dysfunction; fibroblast growth factor receptor 2.
Copyright © 2017 the American Physiological Society.
Figures


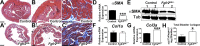



References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

