Pericytes and immune cells contribute to complement activation in tubulointerstitial fibrosis
- PMID: 28052876
- PMCID: PMC5374314
- DOI: 10.1152/ajprenal.00604.2016
Pericytes and immune cells contribute to complement activation in tubulointerstitial fibrosis
Abstract
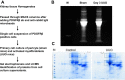
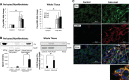
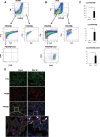
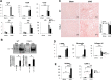
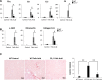
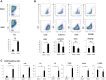
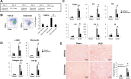
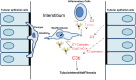
We have examined the pathogenic role of increased complement expression and activation during kidney fibrosis. Here, we show that PDGFRβ-positive pericytes isolated from mice subjected to obstructive or folic acid injury secrete C1q. This was associated with increased production of proinflammatory cytokines, extracellular matrix components, collagens, and increased Wnt3a-mediated activation of Wnt/β-catenin signaling, which are hallmarks of myofibroblast activation. Real-time PCR, immunoblots, immunohistochemistry, and flow cytometry analysis performed in whole kidney tissue confirmed increased expression of C1q, C1r, and C1s as well as complement activation, which is measured as increased synthesis of C3 fragments predominantly in the interstitial compartment. Flow studies localized increased C1q expression to PDGFRβ-positive pericytes as well as to CD45-positive cells. Although deletion of C1qA did not prevent kidney fibrosis, global deletion of C3 reduced macrophage infiltration, reduced synthesis of C3 fragments, and reduced fibrosis. Clodronate mediated depletion of CD11bF4/80 high macrophages in UUO mice also reduced complement gene expression and reduced fibrosis. Our studies demonstrate local synthesis of complement by both PDGFRβ-positive pericytes and CD45-positive cells in kidney fibrosis. Inhibition of complement activation represents a novel therapeutic target to ameliorate fibrosis and progression of chronic kidney disease.
Keywords: C1q; C3; pericytes.
Figures



References
-
- Boor P, Konieczny A, Villa L, Schult AL, Bücher E, Rong S, Kunter U, van Roeyen CR, Polakowski T, Hawlisch H, Hillebrandt S, Lammert F, Eitner F, Floege J, Ostendorf T. Complement C5 mediates experimental tubulointerstitial fibrosis. J Am Soc Nephrol 18: 1508–1515, 2007. doi: 10.1681/ASN.2006121343. - DOI - PubMed
-
- Bossi F, Tripodo C, Rizzi L, Bulla R, Agostinis C, Guarnotta C, Munaut C, Baldassarre G, Papa G, Zorzet S, Ghebrehiwet B, Ling GS, Botto M, Tedesco F. C1q as a unique player in angiogenesis with therapeutic implication in wound healing. Proc Natl Acad Sci U S A 111: 4209–4214, 2014. doi: 10.1073/pnas.1311968111. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

