Orientation selectivity in the visual cortex of the nine-banded armadillo
- PMID: 28053246
- PMCID: PMC5357719
- DOI: 10.1152/jn.00851.2016
Orientation selectivity in the visual cortex of the nine-banded armadillo
Abstract
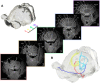
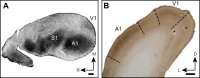
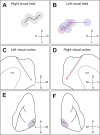
Orientation selectivity in primary visual cortex (V1) has been proposed to reflect a canonical computation performed by the neocortical circuitry. Although orientation selectivity has been reported in all mammals examined to date, the degree of selectivity and the functional organization of selectivity vary across mammalian clades. The differences in degree of orientation selectivity are large, from reports in marsupials that only a small subset of neurons are selective to studies in carnivores, in which it is rare to find a neuron lacking selectivity. Furthermore, the functional organization in cortex varies in that the primate and carnivore V1 is characterized by an organization in which nearby neurons share orientation preference while other mammals such as rodents and lagomorphs either lack or have only extremely weak clustering. To gain insight into the evolutionary emergence of orientation selectivity, we examined the nine-banded armadillo, a species within the early placental clade Xenarthra. Here we use a combination of neuroimaging, histological, and electrophysiological methods to identify the retinofugal pathways, locate V1, and for the first time examine the functional properties of V1 neurons in the armadillo (Dasypus novemcinctus) V1. Individual neurons were strongly sensitive to the orientation and often the direction of drifting gratings. We uncovered a wide range of orientation preferences but found a bias for horizontal gratings. The presence of strong orientation selectivity in armadillos suggests that the circuitry responsible for this computation is common to all placental mammals.NEW & NOTEWORTHY The current study shows that armadillo primary visual cortex (V1) neurons share the signature properties of V1 neurons of primates, carnivorans, and rodents. Furthermore, these neurons exhibit a degree of selectivity for stimulus orientation and motion direction similar to that found in primate V1. Our findings in armadillo visual cortex suggest that the functional properties of V1 neurons emerged early in the mammalian lineage, near the time of the divergence of marsupials.
Keywords: extracellular recording; lateral geniculate nucleus; primary visual cortex.
Copyright © 2017 the American Physiological Society.
Figures


Similar articles
-
Functional cell classes and functional architecture in the early visual system of a highly visual rodent.Prog Brain Res. 2005;149:127-45. doi: 10.1016/S0079-6123(05)49010-X. Prog Brain Res. 2005. PMID: 16226581 Review.
-
Emergence of orientation selectivity in the Mammalian visual pathway.J Neurosci. 2013 Jun 26;33(26):10616-24. doi: 10.1523/JNEUROSCI.0404-13.2013. J Neurosci. 2013. PMID: 23804085 Free PMC article.
-
Mechanisms of Orientation Selectivity in the Primary Visual Cortex.Annu Rev Vis Sci. 2016 Oct 14;2:85-107. doi: 10.1146/annurev-vision-111815-114456. Annu Rev Vis Sci. 2016. PMID: 28532362 Review.
-
Weak orientation and direction selectivity in lateral geniculate nucleus representing central vision in the gray squirrel Sciurus carolinensis.J Neurophysiol. 2015 Apr 1;113(7):2987-97. doi: 10.1152/jn.00516.2014. Epub 2015 Feb 25. J Neurophysiol. 2015. PMID: 25717157 Free PMC article.
-
Functional characterization and spatial clustering of visual cortical neurons in the predatory grasshopper mouse Onychomys arenicola.J Neurophysiol. 2017 Mar 1;117(3):910-918. doi: 10.1152/jn.00779.2016. Epub 2016 Dec 7. J Neurophysiol. 2017. PMID: 27927787 Free PMC article.
Cited by
-
Distribution and diversity of intrinsically photosensitive retinal ganglion cells in tree shrew.J Comp Neurol. 2019 Jan 1;527(1):328-344. doi: 10.1002/cne.24377. Epub 2017 Dec 26. J Comp Neurol. 2019. PMID: 29238991 Free PMC article.
-
Auditory brainstem responses in the nine-banded armadillo (Dasypus novemcinctus).PeerJ. 2023 Dec 13;11:e16602. doi: 10.7717/peerj.16602. eCollection 2023. PeerJ. 2023. PMID: 38107579 Free PMC article.
-
Suppression of top-down influence decreases both behavioral and V1 neuronal response sensitivity to stimulus orientations in cats.Front Behav Neurosci. 2023 Feb 8;17:1061980. doi: 10.3389/fnbeh.2023.1061980. eCollection 2023. Front Behav Neurosci. 2023. PMID: 36844652 Free PMC article.
-
Unsupervised Temporal Contiguity Experience Does Not Break the Invariance of Orientation Selectivity Across Spatial Frequency.Front Syst Neurosci. 2019 May 28;13:22. doi: 10.3389/fnsys.2019.00022. eCollection 2019. Front Syst Neurosci. 2019. PMID: 31231196 Free PMC article.
-
Comparative Analysis of Sectional Anatomy, Computed Tomography and Magnetic Resonance of the Cadaveric Six-Banded Armadillo (Euphractus sexcintus) Head.Vet Sci. 2025 May 1;12(5):433. doi: 10.3390/vetsci12050433. Vet Sci. 2025. PMID: 40431527 Free PMC article.
References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

