Augmenting light coverage for photosynthesis through YFP-enhanced charge separation at the Rhodobacter sphaeroides reaction centre
- PMID: 28054547
- PMCID: PMC5512671
- DOI: 10.1038/ncomms13972
Augmenting light coverage for photosynthesis through YFP-enhanced charge separation at the Rhodobacter sphaeroides reaction centre
Abstract
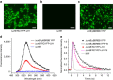
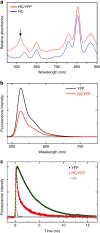
Photosynthesis uses a limited range of the solar spectrum, so enhancing spectral coverage could improve the efficiency of light capture. Here, we show that a hybrid reaction centre (RC)/yellow fluorescent protein (YFP) complex accelerates photosynthetic growth in the bacterium Rhodobacter sphaeroides. The structure of the RC/YFP-light-harvesting 1 (LH1) complex shows the position of YFP attachment to the RC-H subunit, on the cytoplasmic side of the RC complex. Fluorescence lifetime microscopy of whole cells and ultrafast transient absorption spectroscopy of purified RC/YFP complexes show that the YFP-RC intermolecular distance and spectral overlap between the emission of YFP and the visible-region (QX) absorption bands of the RC allow energy transfer via a Förster mechanism, with an efficiency of 40±10%. This proof-of-principle study demonstrates the feasibility of increasing spectral coverage for harvesting light using non-native genetically-encoded light-absorbers, thereby augmenting energy transfer and trapping in photosynthesis.
Conflict of interest statement
The authors declare no competing financial interests.
Figures






References
-
- Blankenship R. E. et al.. Comparing photosynthetic and photovoltaic efficiencies and recognizing the potential for improvement. Science 332, 805–809 (2011). - PubMed
-
- Gundlach K., Werwie M., Wiegand S. & Paulsen H. Filling the “green gap” of the major light-harvesting chlorophyll a/b complex by covalent attachment of rhodamine red. Biochim. Biophys. Acta 1787, 1499–1504 (2009). - PubMed
-
- Reddy K. R. et al.. Palette of lipophilic bacteriochlorins for construction of biohybrid light-harvesting architectures. Chem. Sci. 4, 2036–2053 (2013).
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

