The ultrastructural organization of actin and myosin II filaments in the contractile ring: new support for an old model of cytokinesis
- PMID: 28057763
- PMCID: PMC5328620
- DOI: 10.1091/mbc.E16-06-0466
The ultrastructural organization of actin and myosin II filaments in the contractile ring: new support for an old model of cytokinesis
Abstract
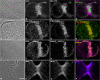
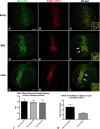
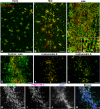
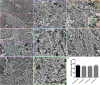
Despite recent advances in our understanding of the components and spatial regulation of the contractile ring (CR), the precise ultrastructure of actin and myosin II within the animal cell CR remains an unanswered question. We used superresolution light microscopy and platinum replica transmission electron microscopy (TEM) to determine the structural organization of actin and myosin II in isolated cortical cytoskeletons prepared from dividing sea urchin embryos. Three-dimensional structured illumination microscopy indicated that within the CR, actin and myosin II filaments were organized into tightly packed linear arrays oriented along the axis of constriction and restricted to a narrow zone within the furrow. In contrast, myosin II filaments in earlier stages of cytokinesis were organized into small, discrete, and regularly spaced clusters. TEM showed that actin within the CR formed a dense and anisotropic array of elongate, antiparallel filaments, whereas myosin II was organized into laterally associated, head-to-head filament chains highly reminiscent of mammalian cell stress fibers. Together these results not only support the canonical "purse-string" model for contractile ring constriction, but also suggest that the CR may be derived from foci of myosin II filaments in a manner similar to what has been demonstrated in fission yeast.
© 2017 Henson et al. This article is distributed by The American Society for Cell Biology under license from the author(s). Two months after publication it is available to the public under an Attribution–Noncommercial–Share Alike 3.0 Unported Creative Commons License (http://creativecommons.org/licenses/by-nc-sa/3.0).
Figures



References
-
- Bonder EM, Fishkind DJ, Henson JH, Cotran NM, Begg DA. Actin in cytokinesis: formation of the contractile apparatus. Zool Sci. 1988;5:699–711.
-
- Boudanoud A, Burian A, Borowska-Wykre˛t D, Uyttewaal M, Wrzalik R, Kwiatkowska D, Hamant O. FibrilTool, an ImageJ plug-in to quantify fibrillar structures in raw microscopy images. Nat Protoc. 2014;9:457–463. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

